

Medical robotics has transformed surgery through precision, automation, and AI, reshaping clinical practice while raising new ethical, economic, and professional challenges.

By Matthew A. McIntosh
Public Historian
Brewminate
Introduction: Machines Enter the Healing Arts
The introduction of robotics into medical care represents one of the most consequential technological shifts in the modern history of medicine. For most of human history, healing was defined by the trained hand, guided by experience, anatomical knowledge, and evolving scientific understanding. The late twentieth century, however, witnessed the emergence of a new paradigm in which machines began to mediate, enhance, and in some cases partially execute clinical interventions. This transition did not simply add new tools to the physician’s repertoire; it fundamentally altered the relationship between practitioner, patient, and procedure, raising new questions about precision, control, and the nature of medical expertise.
Medical robotics emerged at the intersection of several technological trajectories, including advances in computing, imaging, and industrial automation. By the 1980s, robotic systems had already transformed manufacturing through their capacity for repetitive precision and programmable control, particularly in environments where consistency and efficiency were paramount. These systems demonstrated that machines could perform complex, delicate tasks with a level of stability that exceeded human endurance, operating without fatigue and with minimal deviation from programmed parameters. Parallel developments in medical imaging, such as computed tomography (CT) and magnetic resonance imaging (MRI), provided increasingly detailed views of the human body, enabling clinicians to conceptualize surgical intervention in more precise spatial terms. Researchers and clinicians began to recognize that these technological advances could be combined, allowing robotic systems to operate with guidance derived from sophisticated imaging data. The application of robotics to medicine was not an isolated innovation but part of a broader movement toward technological mediation in complex human activities, in which machines extended the limits of human capability while remaining under human direction and control.
The first clinical uses of robotic systems in medicine, particularly the adaptation of industrial robotic arms such as the PUMA 560, demonstrated both the promise and the uncertainty of this new approach. Early procedures, including neurosurgical biopsies, highlighted the potential for enhanced precision and reduced invasiveness, while also revealing the challenges of integrating mechanical systems into delicate biological contexts. These initial experiments did not immediately establish robotics as a standard component of medical practice, but they marked a critical proof of concept. They showed that machines could operate within the highly constrained and sensitive environment of the human body with a level of control that complemented, rather than supplanted, the surgeon’s expertise.
The history of robotics in medical care is best understood not as a sudden technological revolution, but as a gradual process of integration and transformation. From its origins in industrial automation to its current role in minimally invasive surgery and AI-assisted procedures, medical robotics reflects a broader reconfiguration of how care is conceptualized and delivered. By tracing this development across key milestones and contextualizing it within larger historical and technological trends, it becomes possible to see robotics not merely as a tool, but as a defining feature of contemporary medicine’s ongoing evolution toward precision, standardization, and technologically mediated intervention.
Pre-Robotic Foundations: Automation, Cybernetics, and Surgical Precision

The emergence of medical robotics cannot be understood without first examining the intellectual and technological foundations that preceded it. Long before robots entered the operating room, the twentieth century witnessed a profound transformation in how machines were conceived as extensions of human capability. Automation, initially developed to improve efficiency and consistency in industrial production, introduced the idea that complex physical tasks could be delegated to mechanical systems governed by precise control mechanisms. This shift laid the groundwork for later applications in medicine, where precision and repeatability would prove equally valuable.
A central intellectual influence in this transformation was the development of cybernetics, particularly through the work of Norbert Wiener. Cybernetics focused on systems of control and communication in both machines and living organisms, emphasizing feedback loops, regulation, and adaptation. These concepts resonated strongly with medical thinking, especially in physiology, where the human body was increasingly understood as a system of interdependent processes that maintained equilibrium through constant adjustment. Wiener’s framework encouraged scientists to think of both biological organisms and machines in terms of information processing, where signals could be received, interpreted, and acted upon in real time. This perspective helped dissolve rigid boundaries between the mechanical and the organic, opening the door to technologies that could interact dynamically with living systems. In a medical context, this meant imagining devices that could respond to patient-specific conditions rather than simply execute fixed actions, a conceptual leap that would later underpin the development of responsive and semi-autonomous surgical technologies.
Parallel to these theoretical developments, advances in computing technology began to reshape the possibilities of surgical practice. Early computer systems enabled the processing of complex data and the modeling of anatomical structures, allowing for more precise planning of interventions. As computing power increased, so too did the ability to simulate surgical procedures, analyze outcomes, and refine techniques before entering the operating room. By the 1970s and 1980s, the integration of imaging technologies such as CT scanning had transformed diagnostic capabilities, offering detailed, three-dimensional representations of internal anatomy that could be manipulated and studied from multiple angles. These innovations did more than improve diagnosis; they fundamentally altered how surgeons conceptualized the body, shifting from a primarily tactile understanding to one informed by visual and data-driven representations. The combination of computational modeling and advanced imaging created a new paradigm in which surgical precision could be planned and optimized in advance, laying essential groundwork for the later incorporation of robotic systems that could execute these plans with mechanical accuracy.
Industrial robotics further contributed to this evolving landscape by demonstrating the practical capabilities of programmable machines. Robotic arms used in manufacturing environments were capable of executing highly controlled movements with consistent accuracy over extended periods. Unlike human operators, these systems did not suffer from fatigue or variability, making them ideal for tasks requiring exact repetition. Engineers and medical researchers began to explore whether such systems could be adapted for clinical use, particularly in procedures where precision was critical and margins for error were minimal.
Developments in surgical technique were moving toward greater precision and less invasiveness. The rise of microsurgery and minimally invasive procedures reflected a broader shift in medical priorities, emphasizing reduced trauma, faster recovery, and improved patient outcomes. These techniques required enhanced control and visualization, often pushing the limits of what could be achieved by the human hand alone. Surgeons increasingly relied on specialized instruments, magnification technologies, and refined procedural methods to operate within smaller anatomical spaces while minimizing damage to surrounding tissue. This growing demand for precision created both a challenge and an opportunity, as traditional manual techniques approached their practical limits. The potential value of robotic assistance became more apparent, offering the possibility of extending surgical capability beyond natural human dexterity. The convergence of these evolving surgical demands with advances in automation and computation created a fertile environment for the introduction of robotic systems into clinical practice.
These pre-robotic foundations illustrate that the arrival of robotics in medicine was not a sudden innovation but the result of decades of interdisciplinary development. Automation provided the tools, cybernetics offered the conceptual framework, and advances in computing and imaging supplied the necessary data and visualization. By the time robotic systems were first introduced into clinical settings in the 1980s, the intellectual and technological conditions for their success had already been firmly established.
The First Breakthrough: PUMA 560 and Neurosurgical Precision (1985–1987)

The first true breakthrough in medical robotics came with the adaptation of industrial robotic systems for use in clinical settings, most notably the PUMA 560. Originally designed for manufacturing, this robotic arm was repurposed in 1985 to assist in a neurosurgical biopsy, marking the first documented instance of a robot being used in a human surgical procedure. This moment represented more than a technical achievement; it signaled a conceptual shift in how surgery could be performed. Precision, long limited by human steadiness and endurance, could now be augmented by mechanical systems capable of executing movements with extraordinary consistency.
The use of the PUMA 560 in neurosurgery was particularly significant because of the demands of stereotactic procedures, which require exact targeting within the brain. Even minor deviations could have serious consequences, making accuracy paramount. The robotic system, guided by imaging data, was able to position surgical instruments with a level of precision measured in fractions of a millimeter. This demonstrated that robotic assistance could enhance surgical accuracy beyond what was reliably achievable by human hands alone, especially in procedures where spatial constraints and sensitivity were critical.
Following this initial success, researchers explored additional applications for robotic assistance, including its adaptation for laparoscopic surgery. By 1987, the PUMA system had been used in experimental contexts to assist with procedures such as gallbladder removal, extending its relevance beyond neurosurgery. These early efforts highlighted both the flexibility of robotic systems and the challenges of integrating them into different surgical environments. Unlike the controlled conditions of industrial settings, the human body presented variability, complexity, and unpredictability that required careful calibration and oversight. Surgeons had to account for factors such as tissue elasticity, patient movement, and differences in anatomical structure, all of which complicated the direct translation of industrial precision into clinical practice. This required the development of new interfaces, improved imaging integration, and more refined control systems that could accommodate the dynamic conditions of surgery. The process of adaptation was not merely technical but conceptual, forcing practitioners and engineers to rethink how robotic systems could function effectively within living systems rather than static production lines.
Despite its promise, the PUMA 560 remained largely experimental in its medical applications. Limitations in computing power, interface design, and real-time feedback constrained its broader adoption. Surgeons still relied heavily on their own judgment and manual skill, with the robot serving as a highly specialized tool rather than an autonomous system. Nevertheless, the successful use of the PUMA arm established a proof of concept that would guide future development. It demonstrated that robotic systems could operate safely within the human body when properly controlled and integrated with imaging technologies.
The legacy of the PUMA 560 lies in its role as a bridge between industrial automation and clinical practice. It did not revolutionize surgery overnight, but it provided the first clear evidence that robotics could meaningfully contribute to medical care. By proving that machines could enhance precision in delicate procedures, it laid the groundwork for the development of more advanced, purpose-built medical robotic systems in the decades that followed.
Specialization and FDA Approval: AESOP and ROBODOC (1990–1992)

The early 1990s marked a decisive transition in the history of medical robotics, as experimental systems began to evolve into specialized tools with defined clinical roles. Unlike the earlier adaptation of industrial machines such as the PUMA 560, this period saw the development of robots designed specifically for medical use. These systems were not merely proofs of concept but were engineered to address particular surgical needs, signaling a shift from exploratory innovation to targeted application. This movement toward specialization reflected a growing confidence in the viability of robotics in clinical environments and a recognition that different procedures required tailored technological solutions.
One of the most significant developments of this period was the introduction of the Automated Endoscopic System for Optimal Positioning (AESOP), created by Computer Motion. AESOP functioned as a robotic camera holder for laparoscopic surgery, allowing surgeons to control the position of the endoscope through voice commands. This innovation addressed a practical limitation in minimally invasive procedures, where maintaining a stable and responsive visual field was essential. By replacing the need for a human assistant to hold and adjust the camera, AESOP improved consistency, reduced fatigue, and allowed surgeons greater autonomy during operations. Its approval by the United States Food and Drug Administration (FDA) in 1994 marked a critical milestone, establishing it as the first widely recognized surgical robot to receive regulatory clearance for clinical use.
At nearly the same time, the development of ROBODOC represented a different approach to medical robotics, focusing on precision in orthopedic surgery. Designed for use in hip arthroplasty, ROBODOC was capable of performing highly accurate bone milling based on preoperative imaging data. This level of precision was particularly important in joint replacement procedures, where even minor deviations could affect implant fit and long-term outcomes. ROBODOC demonstrated that robotic systems could not only assist surgeons but also execute specific tasks with a degree of consistency that improved reproducibility across procedures. Its introduction reinforced the idea that robotics could enhance both the planning and execution phases of surgery.
The emergence of AESOP and ROBODOC also highlighted the importance of regulatory frameworks in shaping the development of medical robotics. FDA approval served not only as a validation of safety and efficacy but also as a gateway to broader clinical adoption. Manufacturers were required to demonstrate that these systems met rigorous standards, encouraging more systematic testing and refinement. Regulatory oversight helped define the boundaries of robotic use, ensuring that these technologies were integrated into medical practice in a controlled and accountable manner. This process contributed to the gradual normalization of robotic assistance in surgery, transforming it from a novel concept into an accepted component of clinical care.
The introduction of AESOP and ROBODOC marked a critical stage in the evolution of medical robotics. These systems demonstrated that specialized, purpose-built robots could address specific surgical challenges with measurable benefits. More importantly, they established a model for future development, combining technological innovation with regulatory approval and clinical validation. By the early 1990s, medical robotics had moved beyond experimentation, entering a phase in which precision, reliability, and institutional acceptance began to define its role in modern medicine.
The Turning Point: The da Vinci System and the Rise of Robotic Surgery (2000)
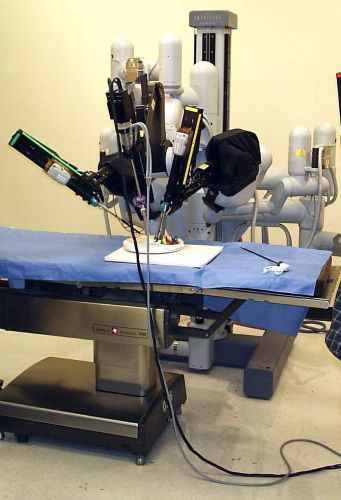
The approval of the da Vinci Surgical System in 2000 marked a decisive turning point in the history of medical robotics, transforming a field of specialized and experimental devices into a widely adopted surgical paradigm. Developed by Intuitive Surgical, the system represented a significant departure from earlier robotic tools such as AESOP and ROBODOC, which were designed for narrowly defined tasks. Instead, the da Vinci platform introduced a versatile, multi-purpose system capable of supporting a wide range of procedures across different medical specialties. Its approval by the United States Food and Drug Administration (FDA) for general laparoscopic surgery signaled not only technological maturity but also institutional confidence in the role of robotics in routine clinical practice.
At the core of the da Vinci system was its teleoperated design, which placed the surgeon at a console rather than directly at the patient’s side. From this position, the surgeon controlled robotic arms equipped with specialized instruments that translated hand movements into precise micro-movements within the body. This setup allowed for a level of dexterity and stability that surpassed traditional laparoscopic techniques, particularly through the use of articulated, “wristed” instruments that mimicked and extended the natural range of motion of the human hand. The system also filtered out tremors and scaled movements, enabling surgeons to perform delicate tasks with enhanced control.
Equally important was the integration of advanced visualization technologies. The da Vinci system provided high-definition, three-dimensional imaging of the surgical field, offering depth perception that had been largely absent in earlier minimally invasive procedures. This improvement significantly enhanced the surgeon’s ability to navigate complex anatomical structures, reducing the cognitive and physical strain associated with traditional laparoscopy. The immersive visual environment allowed for more precise identification of tissue planes, vascular structures, and subtle anatomical variations that might otherwise be difficult to discern. In addition, the system’s camera stability and magnification capabilities enabled a level of visual consistency that improved both accuracy and efficiency during procedures. The combination of enhanced visualization and mechanical precision created a new standard for surgical performance, particularly in procedures requiring fine manipulation and careful dissection.
The versatility of the da Vinci system contributed to its rapid adoption across multiple specialties, including urology, cardiothoracic surgery, gynecology, and general surgery. Procedures such as prostatectomies became closely associated with robotic assistance, as surgeons recognized the benefits of improved precision and reduced invasiveness. Hospitals and surgical centers began investing in robotic systems not only for their clinical advantages but also for their potential to attract patients seeking advanced treatment options. Robotic surgery quickly transitioned from a novel innovation to a competitive standard within modern healthcare systems.
The widespread adoption of the da Vinci system also introduced new challenges and debates. The high cost of acquisition and maintenance raised concerns about accessibility and the potential for disparities in care between institutions with differing resources. In many cases, the financial burden of acquiring and maintaining robotic systems was passed on indirectly through healthcare costs, prompting questions about cost-effectiveness and value. Additionally, the reliance on complex technology required significant training and adaptation for surgeons, leading to discussions about learning curves and the standardization of robotic surgical education. Surgeons needed to develop new forms of hand-eye coordination mediated through digital interfaces, as well as an understanding of system limitations and troubleshooting. Critics also questioned whether the benefits of robotic surgery consistently justified its costs, particularly in procedures where traditional methods already yielded strong outcomes, fueling ongoing debates in both clinical and policy circles.
Despite these challenges, the da Vinci system fundamentally reshaped the trajectory of medical robotics. It demonstrated that robotic platforms could be integrated into everyday surgical practice at scale, setting a precedent for future innovations. More than any previous system, it established robotics as a central component of modern medicine, redefining expectations for precision, visualization, and minimally invasive care. It marked the transition from isolated technological advancement to a sustained transformation in how surgery is performed.
From Specialized Tools to Multipurpose Systems (2000s Expansion)
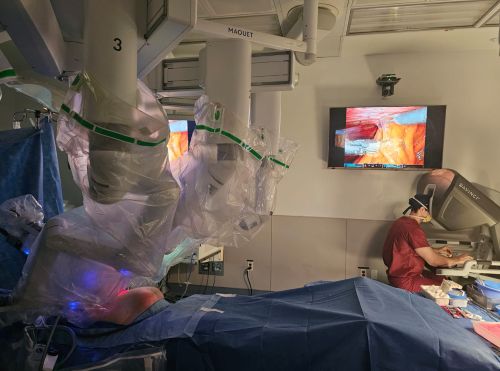
The early 2000s marked a period of rapid expansion in medical robotics, building directly on the foundation established by the da Vinci system. What had once been a collection of specialized devices designed for narrow applications began to evolve into flexible, multipurpose platforms capable of supporting a wide range of surgical procedures. This shift reflected both technological advancement and growing institutional confidence, as hospitals increasingly recognized robotics as a long-term investment rather than an experimental addition. The focus moved away from isolated innovations toward integrated systems that could be deployed across multiple departments and specialties.
One of the defining characteristics of this period was the increasing versatility of robotic platforms. Systems were refined to accommodate diverse surgical needs, allowing a single platform to be used in urology, gynecology, general surgery, and cardiothoracic procedures. This adaptability was made possible by modular instrument design, improved software interfaces, and enhanced imaging integration. Surgeons could tailor the system to specific procedures while maintaining a consistent operational framework, reducing the need for entirely different technologies for each type of surgery. Robotics began to function less as a specialized tool and more as a standard surgical infrastructure.
The expansion of robotic systems was closely tied to the growth of minimally invasive surgical techniques. As patient demand for shorter recovery times and reduced postoperative complications increased, hospitals sought technologies that could support these outcomes. Robotic systems offered improved precision, smaller incisions, and enhanced control, making them particularly well suited to minimally invasive approaches. Clinical studies during this period began to demonstrate measurable benefits in certain procedures, including reduced blood loss, shorter hospital stays, and faster recovery times for specific operations such as prostatectomy and hysterectomy. These benefits were not uniform across all procedures, and outcomes often depended on surgeon experience and institutional expertise. This variability contributed to ongoing debates about when robotic intervention provided clear advantages over established laparoscopic techniques. Nevertheless, the alignment between robotic capability and the goals of minimally invasive surgery reinforced the broader adoption of these systems.
The adoption of robotic surgery required significant changes in surgical training and professional practice. Surgeons had to learn new techniques that differed fundamentally from traditional open or laparoscopic methods, including operating from a console and interpreting three-dimensional visual data. Training programs emerged to address these needs, often incorporating simulation-based learning and credentialing processes to ensure proficiency. This transformation in training underscored the extent to which robotics was not simply an addition to existing practice but a redefinition of surgical skill itself.
Economic considerations also played a central role in the expansion of medical robotics during the 2000s. The cost of acquiring and maintaining robotic systems remained substantial, leading to debates about cost-effectiveness and resource allocation. Hospitals had to weigh the potential clinical benefits against financial constraints, particularly in healthcare systems with limited funding. The presence of advanced robotic technology became a marker of institutional prestige, influencing patient choice and competitive positioning. This dynamic contributed to the continued spread of robotic systems despite ongoing concerns about cost.
By the end of the decade, medical robotics had firmly established itself as a central component of modern surgical practice. The transition from specialized tools to multipurpose systems reflected a broader shift toward integration, standardization, and scalability. Robotics was no longer confined to experimental or highly specialized contexts but had become embedded in the everyday functioning of many healthcare institutions. This normalization was evident not only in the number of procedures performed but also in the expectations of both patients and practitioners, who increasingly viewed robotic assistance as a standard option rather than an exceptional one. As training programs expanded and technologies improved, the barriers to adoption gradually decreased, further accelerating integration. This period of expansion set the stage for subsequent innovations, including the incorporation of artificial intelligence, enhanced imaging systems, and robotic-assisted diagnostics, all of which would continue to reshape the field in the years to come.
Contemporary Developments: AI Integration and Precision Medicine
The most recent phase in the evolution of medical robotics has been defined by the integration of artificial intelligence, advanced imaging, and data-driven decision-making into surgical systems. While earlier generations of robots emphasized mechanical precision and stability, contemporary systems increasingly incorporate computational intelligence that enhances planning, execution, and evaluation. This shift reflects a broader transformation in medicine toward precision and personalization, where treatments are tailored to individual patients based on detailed data analysis and predictive modeling. The convergence of robotics and artificial intelligence has allowed for a more holistic approach to care, in which diagnosis, planning, and intervention are interconnected within a single technological framework. Robotics has become not only a tool for physical intervention but also a platform for integrating complex streams of medical information into clinical practice, enabling a more coordinated and data-informed approach to treatment.
One of the most significant developments has been the use of artificial intelligence to assist in surgical planning and intraoperative guidance. Machine learning algorithms can analyze large datasets, including imaging scans and patient histories, to identify patterns and recommend optimal approaches to treatment. In some systems, AI is used to map surgical pathways, anticipate potential complications, and provide real-time feedback during procedures. This capability allows surgeons to make more informed decisions, supported by data that would be difficult to process manually. Although these systems do not replace human judgment, they augment it, offering a level of analytical support that enhances both accuracy and efficiency.
Advances in imaging technology have further strengthened the relationship between robotics and precision medicine. High-resolution imaging, combined with real-time tracking, enables surgeons to visualize anatomical structures with unprecedented clarity. Robotic systems can integrate this imaging data directly into their operational framework, allowing for more precise targeting and manipulation of tissues. In orthopedic surgery, for example, robotic-arm assisted systems such as MAKO use preoperative imaging to create detailed models of the patient’s anatomy, guiding the placement of implants with a high degree of accuracy. These developments illustrate how robotics and imaging have become increasingly interdependent, each enhancing the capabilities of the other.
The growing role of data in medical robotics has introduced new possibilities and challenges. The collection and analysis of surgical data allow for continuous improvement in techniques and outcomes, as systems can be refined based on accumulated experience. This data-driven approach supports the development of standardized procedures while also enabling customization for individual patients. It also raises important questions about data privacy, security, and the ethical use of patient information. As robotic systems become more interconnected and reliant on digital infrastructure, ensuring the integrity and protection of medical data has become a critical concern.
The integration of artificial intelligence and advanced technologies into medical robotics represents a significant step toward the future of healthcare. These systems are not yet fully autonomous, but they are increasingly capable of supporting complex decision-making and enhancing surgical precision. As research and development continue, the boundaries between human expertise and machine assistance are likely to become more fluid, leading to new forms of collaboration between surgeons and technology. In this evolving landscape, medical robotics stands at the forefront of a broader transformation in medicine, one that emphasizes precision, adaptability, and the intelligent use of data.
Patient Outcomes and the Rise of Minimally Invasive Surgery
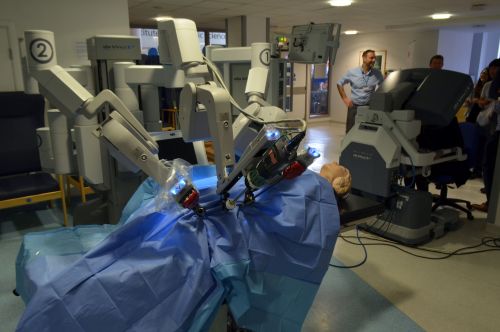
The expansion of medical robotics has been closely linked to the broader rise of minimally invasive surgery, a development that has fundamentally reshaped patient care in the late twentieth and early twenty-first centuries. Unlike traditional open surgery, which often requires large incisions and extended recovery periods, minimally invasive techniques aim to reduce physical trauma while maintaining or improving clinical effectiveness. Robotic systems have played a crucial role in advancing this approach by enhancing precision, stability, and visualization, allowing surgeons to operate through smaller access points with greater control. The adoption of robotics has been driven not only by technological innovation but also by a growing emphasis on improving patient outcomes.
One of the most significant benefits associated with robotic-assisted minimally invasive surgery is the reduction in recovery time. Patients undergoing these procedures often experience shorter hospital stays, less postoperative pain, and a quicker return to normal activities. These advantages are particularly evident in procedures such as prostatectomy, hysterectomy, and certain cardiac surgeries, where robotic systems enable more precise dissection and reduced disruption of surrounding tissues. The cumulative effect of these improvements has been a shift in patient expectations, with minimally invasive options increasingly viewed as the preferred standard when available.
In addition to faster recovery, robotic-assisted surgery has been associated with lower rates of certain complications. Enhanced precision can reduce the likelihood of unintended damage to adjacent structures, while improved visualization allows surgeons to navigate complex anatomical regions more effectively. Studies have shown reductions in blood loss and infection rates in specific procedures, although these outcomes are not universal and depend on factors such as surgeon experience, case complexity, and institutional expertise. In some instances, robotic systems have enabled surgeons to perform procedures that would otherwise be more difficult or risky using conventional techniques, particularly in anatomically constrained or delicate areas. The introduction of new technology has brought its own risks, including system malfunctions or technical limitations that must be carefully managed. The variability in results has prompted ongoing research into the conditions under which robotic systems provide the greatest clinical benefit, as well as continued efforts to refine both technology and training to ensure consistent outcomes across different settings.
The rise of minimally invasive robotic surgery has introduced new considerations regarding access and equity. While patients treated at well-funded institutions may benefit from the latest technologies, those in resource-limited settings may not have the same opportunities. The cost of robotic systems, combined with the need for specialized training and infrastructure, has contributed to disparities in availability. This uneven distribution raises important questions about how innovations in medical technology can be implemented in ways that promote broader access to improved care.
Ultimately, the relationship between robotics and patient outcomes reflects a complex interplay between technological capability, clinical practice, and healthcare systems. While robotic-assisted minimally invasive surgery has demonstrated clear advantages in many contexts, its benefits are not uniform across all procedures or patient populations. Nevertheless, its continued development and adoption underscore a broader trend in medicine toward reducing invasiveness while enhancing precision. Robotics has not only improved specific surgical outcomes but has also contributed to a larger redefinition of what patients and practitioners expect from modern medical care.
Ethical, Economic, and Professional Implications
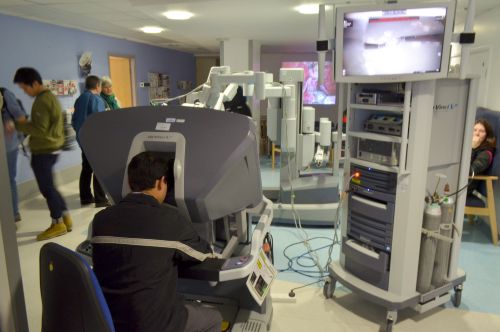
The rapid integration of robotic systems into surgical practice has introduced a complex web of ethical considerations that extend beyond technical performance. At the center lies the question of responsibility. When outcomes depend on a combination of human decision-making and machine execution, assigning accountability becomes less straightforward. Surgeons remain legally and ethically responsible, yet their reliance on proprietary systems complicates notions of autonomy and control. This tension reflects broader debates in the philosophy of technology regarding human agency in increasingly mediated environments.
Economic factors further complicate the landscape. Robotic surgical systems, particularly platforms like the da Vinci system, require substantial capital investment, ongoing maintenance costs, and specialized training. These expenses can exacerbate disparities between well-funded institutions and under-resourced hospitals, raising concerns about equitable access to advanced care. While proponents argue that improved outcomes and reduced recovery times may offset initial costs, the evidence remains uneven, particularly when examined across diverse healthcare systems and patient populations.
The professional identity of surgeons has also undergone subtle but significant transformation. Mastery of robotic interfaces requires a different skill set than traditional open or even laparoscopic surgery, emphasizing console-based dexterity, spatial interpretation through screens, and reliance on digital feedback systems. This shift has prompted debates within the surgical community about what constitutes expertise, as well as concerns about deskilling in certain manual techniques. Some critics argue that an overreliance on robotic systems may erode tactile familiarity with human anatomy, a skill historically central to surgical judgment. Others counter that robotic platforms enhance precision and reduce fatigue, ultimately expanding what surgeons are capable of achieving. New forms of specialization have emerged, with surgeons increasingly defined by their proficiency with specific platforms rather than by anatomical or procedural domains alone, signaling a broader reconfiguration of professional identity in technologically mediated medicine.
Training pathways have adapted in response to these changes, but not without friction. Residency programs must balance the need to teach foundational surgical skills with the demands of emerging technologies. Simulation-based training has become more prominent, allowing trainees to develop competence in robotic systems before entering the operating room. Access to such training tools is not uniform, potentially reinforcing existing institutional hierarchies within medical education. The question of how best to certify and credential robotic proficiency remains an evolving challenge.
Ethical concerns also extend to patient consent and understanding. While robotic surgery is often marketed as cutting-edge and minimally invasive, patients may not fully grasp the role of the machine versus the surgeon. Ensuring informed consent requires transparent communication about risks, benefits, and the nature of the technology involved. This is particularly important given that some studies suggest outcomes may depend more on surgeon experience than on the technology itself, complicating simplistic narratives of technological superiority. This means that patient expectations can be shaped as much by marketing language as by clinical realities, raising concerns about whether consent is truly informed or subtly influenced. Moreover, disparities in health literacy can further complicate this process, leaving some patients less equipped to critically evaluate their options. As robotic systems become more normalized, the ethical obligation to clearly articulate their role only becomes more pressing, especially in environments where institutional prestige and technological branding intersect with patient trust.
Finally, the increasing involvement of corporate entities in the development and dissemination of surgical robots introduces additional layers of influence. Intellectual property protections, proprietary software, and market competition shape not only the availability of these systems but also the research that evaluates them. This raises important questions about the independence of clinical evidence and the potential for commercial interests to shape medical practice. As robotic surgery continues to evolve, these ethical, economic, and professional dimensions will remain central to understanding its place within modern healthcare.
Historiography: Interpreting the Rise of Medical Robotics
Historians of medicine have approached the rise of medical robotics through multiple interpretive frameworks, reflecting broader debates about technology, agency, and progress. Early accounts tended to emphasize innovation as a linear and largely beneficial process, portraying robotic systems as the natural culmination of centuries of surgical refinement. In this view, the emergence of robotic platforms represented not a rupture but a continuation of longstanding efforts to enhance precision, reduce human error, and extend the capabilities of the physician. Such narratives often drew implicitly on a Whiggish model of technological development, privileging success stories and framing resistance as temporary or uninformed.
More recent scholarship has complicated this narrative by situating medical robotics within the institutional and economic structures that enabled its development. Rather than viewing these systems as inevitable advancements, historians have examined the roles of military research, corporate investment, and regulatory frameworks in shaping their trajectory. The influence of Cold War funding on early developments in teleoperation and cybernetics, for example, has been highlighted as a critical precondition for later medical applications. This perspective shifts attention away from individual inventors and toward the networks of power and capital that make technological adoption possible, aligning the history of medical robotics with broader trends in the history of science and technology.
A parallel line of interpretation has focused on the cultural meanings attached to robotic surgery, particularly in relation to public perceptions of medicine. Scholars have explored how the language of innovation, precision, and futurity has been mobilized in both professional and commercial contexts to legitimize these systems. Advertising materials, hospital marketing campaigns, and media representations have all contributed to the construction of robotic surgery as a symbol of modernity and superiority. This cultural framing has, in turn, influenced patient expectations and institutional decision-making, suggesting that the adoption of robotic systems cannot be understood solely in terms of clinical efficacy. In many cases, hospitals have leveraged robotic platforms as markers of prestige, using their presence to signal technological leadership regardless of measurable outcome differences. Media narratives have further amplified this perception, often presenting robotic surgery as inherently safer or more advanced, even when comparative data remains inconclusive. The historiography increasingly recognizes that perception, marketing, and symbolic capital play a substantial role in the diffusion of medical robotics, shaping both demand and institutional priorities in ways that extend beyond strictly medical considerations.
Critical historians have raised questions about the extent to which medical robotics represents genuine transformation versus technological reframing of existing practices. Some argue that many robotic procedures replicate techniques developed in laparoscopic surgery, offering incremental rather than revolutionary change. From this perspective, the significance of robotics lies less in its clinical novelty and more in its reorganization of surgical labor, training, and authority. By mediating the surgeon’s interaction with the patient through complex interfaces, robotic systems alter the sensory and cognitive dimensions of surgical practice, even when the underlying procedures remain familiar.
Finally, historiographical debates have increasingly turned toward questions of equity and global distribution. While much of the literature has focused on developments in North America and Europe, scholars are beginning to examine how medical robotics is adopted, adapted, or resisted in other contexts. This includes attention to disparities in access, the role of international markets, and the ways in which local medical cultures shape the reception of new technologies. Historians are moving toward a more global and critical account of medical robotics, one that recognizes both its transformative potential and its embeddedness within unequal systems of power and resource distribution.
Conclusion: Precision, Power, and the Future of Care
The history of medical robotics reveals a pattern that is neither purely revolutionary nor merely incremental, but instead deeply entangled with broader transformations in technology, knowledge, and institutional power. From early experiments in automation and imaging to the widespread adoption of robotic surgical platforms, the trajectory has been shaped as much by cultural expectations and economic incentives as by clinical necessity. What emerges is not a simple story of progress, but a layered evolution in which innovation redefines the boundaries of medical possibility while simultaneously introducing new forms of dependence and complexity.
Precision, often cited as the defining advantage of robotic systems, carries with it its own set of implications. Enhanced accuracy in surgical procedures has undeniably improved outcomes in many contexts, particularly in minimally invasive operations. Yet precision is not a neutral attribute. It is embedded within systems designed, manufactured, and maintained by corporate and institutional actors, raising questions about control, access, and long-term sustainability. As medicine becomes increasingly mediated by advanced technologies, the locus of expertise shifts, and with it, the balance of authority between practitioner, institution, and machine. This redistribution of authority is not merely technical but structural, influencing how decisions are made, who is empowered to make them, and how accountability is ultimately assigned. In environments where proprietary systems dominate, even highly trained surgeons may find their autonomy constrained by software limitations, licensing agreements, or system-specific protocols. Precision becomes inseparable from power, and the benefits it offers must be weighed against the dependencies it creates within modern healthcare systems.
The growing integration of artificial intelligence and data-driven decision-making further complicates this landscape. Robotic systems are no longer limited to executing human commands with mechanical fidelity; they are beginning to incorporate predictive analytics, adaptive learning, and real-time feedback mechanisms. These developments hold the potential to transform surgical practice in profound ways, enabling levels of consistency and insight previously unattainable. They introduce new ethical and epistemological challenges, particularly concerning transparency, accountability, and the interpretability of machine-assisted decisions.
Ultimately, the future of care will be defined not solely by technological capability but by how these tools are integrated into the human dimensions of medicine. Trust, communication, and professional judgment remain central to the patient experience, even as machines assume a greater role in diagnosis and treatment. The history of medical robotics underscores the importance of approaching innovation critically, recognizing both its transformative potential and its limitations. In this balance between precision and power lies the enduring challenge of ensuring that technological advancement serves not only efficiency and expertise, but also equity, humanity, and care. This requires not only technical refinement but also deliberate policy choices, ethical oversight, and sustained attention to disparities in access and outcomes. Without such considerations, the very technologies designed to enhance care risk reinforcing existing inequalities, privileging those institutions and populations already positioned to benefit. The future of medical robotics will depend as much on governance and values as on engineering, demanding a continuous negotiation between what is possible and what is just.
Bibliography
- Aho, Alfred V., and Jeffrey D. Ullman. Foundations of Computer Science. New York: W. H. Freeman, 1992.
- Barbash, Galit I., and Robert J. Glied. “New Technology and Health Care Costs — The Case of Robot-Assisted Surgery.” New England Journal of Medicine 363, no. 8 (2010): 701–704.
- Davies, B. L. “A Review of Robotics in Surgery.” Proceedings of the Institution of Mechanical Engineers, Part H: Journal of Engineering in Medicine 214, no. 1 (2000): 129–140.
- Gawande, Atul. Better: A Surgeon’s Notes on Performance. New York: Metropolitan Books, 2007.
- Haidegger, Tamás. “Autonomy for Surgical Robots: Concepts and Paradigms.” IEEE Transactions on Medical Robotics and Bionics 1, no. 2 (2019): 65–76.
- Herron, David M., and Michael Marohn. “A Consensus Document on Robotic Surgery.” Surgical Endoscopy 22, no. 2 (2008): 313–325.
- Hounsfield, G. N. “Computerized Transverse Axial Scanning (Tomography): Part 1. Description of System.” British Journal of Radiology 46, no. 552 (1973): 1016–1022.
- Howell, Joel D. Technology in the Hospital: Transforming Patient Care in the Early Twentieth Century. Baltimore: Johns Hopkins University Press, 1995.
- Intuitive Surgical. da Vinci Surgical System: Product Overview and Clinical Applications. Sunnyvale, CA: Intuitive Surgical, 2000.
- Jiang, F., Y. Jiang, H. Zhi, Y. Dong, H. Li, S. Ma, Y. Wang, Q. Dong, H. Shen, and Y. Wang. “Artificial Intelligence in Healthcare: Past, Present and Future.” Stroke and Vascular Neurology 2, no. 4 (2017): 230–243.
- Kazanzides, Peter, et al. “An Integrated System for Cementless Hip Replacement.” IEEE Engineering in Medicine and Biology Magazine 14, no. 3 (1995): 307–313.
- Kwoh, Y. S., J. Hou, E. A. Jonckheere, and S. Hayati. “A Robot with Improved Absolute Positioning Accuracy for CT Guided Stereotactic Brain Surgery.” IEEE Transactions on Biomedical Engineering 35, no. 2 (1988): 153–160.
- Lanfranco, Anthony R., Andres E. Castellanos, Jay P. Desai, and William C. Meyers. “Robotic Surgery: A Current Perspective.” Annals of Surgery 239, no. 1 (2004): 14–21.
- Nof, Shimon Y., ed. Handbook of Industrial Robotics. 2nd ed. New York: Wiley, 1985.
- Pickering, Andrew. The Cybernetic Brain: Sketches of Another Future. Chicago: University of Chicago Press, 2010.
- Satava, Richard M. “Surgical Robotics: The Early Chronicles: A Personal Historical Perspective.” Surgical Laparoscopy, Endoscopy & Percutaneous Techniques 12, no. 1 (2002): 6–16.
- Silver, Julie K., David J. Neumayer, and Michael R. Soper. “The Impact of Robotic Surgery on Surgical Training.” Journal of Surgical Education 69, no. 3 (2012): 347–352.
- Taylor, Russell H., Blake Hannaford, and Peter Kazanzides. “Medical Robotics and Computer-Integrated Surgery.” In Springer Handbook of Robotics, edited by Bruno Siciliano and Oussama Khatib, 1657–1684. Berlin: Springer, 2016.
- Topol, Eric. Deep Medicine: How Artificial Intelligence Can Make Healthcare Human Again. New York: Basic Books, 2019.
- United States Food and Drug Administration. “510(k) Premarket Notification Database.” Accessed 2025.
- —-. “Computer-Assisted Surgical Systems.” Last modified 2024.
- Wiener, Norbert. Cybernetics: Or Control and Communication in the Animal and the Machine. Cambridge, MA: MIT Press, 1948.
Originally published by Brewminate, 03.31.2026, under the terms of a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International license.


