By Dr. John A. Cunningham, et.al.
Postdoctoral Researcher
School of Earth Sciences
University of Bristol
Highlights
- • Computer-aided visualization and analysis has revolutionized the study of fossils.
- • Fossils can now be characterized in three dimensions and in unprecedented detail.
- • The resulting digital reconstructions can be used in rigorous functional analyses.
- • Hypotheses regarding the function of extinct organisms can therefore be tested.
Computer-aided visualization and analysis of fossils has revolutionized the study of extinct organisms. Novel techniques allow fossils to be characterized in three dimensions and in unprecedented detail. This has enabled paleontologists to gain important insights into their anatomy, development, and preservation. New protocols allow more objective reconstructions of fossil organisms, including soft tissues, from incomplete remains. The resulting digital reconstructions can be used in functional analyses, rigorously testing long-standing hypotheses regarding the paleobiology of extinct organisms. These approaches are transforming our understanding of long-studied fossil groups, and of the narratives of organismal and ecological evolution that have been built upon them.
A Digital Revolution in Paleontology
Paleontology has a reputation as a dry and dusty discipline, stymied by privileged access to fossil specimens that are interpreted with an eye of faith and used to evidence just-so stories of adaptive evolution. However, in recent years the discipline has been revolutionized by the emergence of powerful methods for the digital visualization and analysis of fossil material. This has included improvements in both computer technology and its availability, and in tomographic techniques, which have made it possible to image a series of 2D sections or slices through a fossil and to use these to make a 3D reconstruction of the specimen. As a consequence of applying these techniques, paleontological studies are often at the forefront of anatomical research. In fact, some fossil species are now better characterized in terms of their anatomy and development than their living counterparts; for example, the fossil sarcopterygian fish, Eusthenopteron foordi, is among the best known of all fishes living and dead as a consequence of the detailed tomographic studies undertaken by Jarvik 1 and 2. Moreover, the functional morphology of fossil organisms can now be objectively evaluated through quantitative functional analyses that enable definitive tests of hitherto untestable hypotheses in (paleo)biology. A sufficient number of fossil species have now been digitally characterized to make it feasible to carry out comparative functional analyses and reveal trends in functional evolution. Furthermore, this proliferation of digital datasets means that widespread sharing and dissemination of 3D data is now possible, providing a potential solution to problems of access to rare fossil material. Computer-aided visualization and analysis is therefore transforming the way that fossils are studied and, in consequence, revealing ever-greater insights into the paleobiology of extinct groups and the evolutionary theories that are based upon them. Here, we review the methodological advances that have made this revolution possible and discuss the types of paleontological research questions that can now be addressed through computational approaches.
Extracting fossil data from rocks
A major problem that has hampered paleontologists since the beginning of the science has been how to extract fossils from their host rock. The conventional approach is to physically remove the rock from the fossil using either a mechanical method, stripping away the enclosing matrix using tools ranging from needles to dentist’s drills [3], or by exploiting the chemical differences between fossils and their host rock, for example, using weak acetic acid to remove limestone (CaCO3) from phosphatic fossils [4]. Alternatively, fossil morphology can be recovered by dissolving the fossil to leave a void in the rock from which a cast can be made [5]. There are, however, a number of pitfalls associated with the isolation of fossils by these techniques. For instance, they can damage delicate structures, and the targeted extraction of particular features is not straightforward. In some instances, exceptionally preserved soft tissues have been prepared away unwittingly to expose the bone or shell – yet, in retrospect, the soft tissues were the most significant fossil remains. Furthermore, these techniques do not allow the study of internal anatomy. A solution to these problems is to take a tomographic approach, creating a 3D model of the fossil from a series of 2D slices. This powerful methodology enables the anatomy of fossils to be characterized in unprecedented detail, unlocking their potential for enhancing our understanding of the history of life.
Characterizing fossils in 3D
Tomography is not a recent innovation in paleontology. Its roots can be traced to the early twentieth century, when the geological polymath William Sollas began the onerous task of manually grinding away fossils embedded in rock, stopping at regular intervals to photograph or trace by hand the exposed surfaces (Figure 1A, D; [6]). This process of serial grinding tomography produces 2D slices that are used to create physical models of specimens out of wax, cardboard, or polystyrene, thereby visualizing their structure in 3D (Figure 1B, E, F) 6 and 7. This process is extremely time-consuming. For example, it infamously took the Swedish paleontologist Erik Jarvik some 25 years to produce a 3D wax model of the Devonian fish Eusthenopteron based on over 500 detailed drawings [2], after which time the original specimen had been destroyed entirely. Thus, although the serial grinding approach has been applied with considerable success 1, 2, 6, 7, 8, 9 and 10, the degree of time and effort required to image a fossil, as well as, critically, the destructive nature of the method, have limited its widespread adoption. Variants on the approach, for example, using serial sawing to expose sections [11], acetate peels to record the exposed surfaces 12 and 13, or tungsten microtomy to produce very thin sections that can be conserved on glass plates [14], enable the retention of some original material, but the process remains incredibly laborious. Despite these drawbacks, serial grinding tomography is still in use today and has been vastly improved by the integration of digital photography and computer reconstruction, allowing the fossil to be studied and dissected virtually (Box 1).
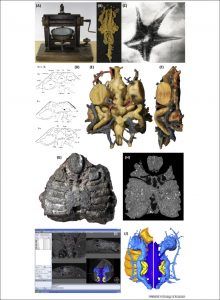
Figure 1: Three-dimensional reconstruction of fossils. (A) The grinding machine used by Sollas in the first paleontological tomographic studies. (B) Sollas’ wax model reconstruction of the early vertebrate Palaeospondylus (specimen length 19 mm). (C) X-Ray image of the asteroid Palaeostella solida from the Hunsrück Slate (specimen diameter 83 mm). (D) Drawings of successive grinding surfaces from the jawless vertebrate Cephalaspis by Stensiö [7] (specimen length 13 mm). (E, F) Stensiö’s reconstruction of Cephalaspis in dorsal (E) and lateral (F) views. (G) Photograph of the jawless galeaspid vertebrate Shuyu (specimen width 10 mm). (H) A synchrotron-based X-ray slice through Shuyu. (I) Screen capture of Shuyu being digitally reconstructed using the software Amira. (J) Digital reconstructions of the right side of the braincase of Shuyu in dorsal (left) and ventral (right) aspect [101].
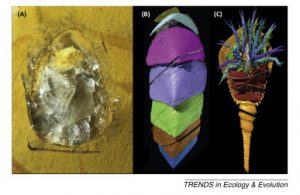
Box 1 – The Herefordshire Lagerstätte: Serial grinding techniques have been used to great effect in the study of the fossils from an important Silurian site of exceptional preservation known as the Herefordshire Lagerstätte 73, 74 and 75. The fossils from this locality are preserved in calcite and can be visually distinguished from the nodules containing them (Figure IA), but have a very similar chemical composition to the host rock which means that they cannot be successfully extracted using physical or chemical preparation methods. Furthermore, X-ray techniques (described in the main text) – which rely on internal density differences – have so far failed to differentiate the fossils from the matrix. Therefore, serial grinding tomography is currently the only way the specimens from this important site can be studied in three dimensions (Figure IB, C). This has provided key insights into the paleobiology and evolution of a range of extinct invertebrate taxa that are unique to this site. Although this type of serial grinding approach works well for most three-dimensionally preserved fossils, the fact that it results in the complete destruction of the specimen means that today it should be seen as a last resort for studying material, such as the fossils from the Herefordshire Lagerstätte, that is not amenable to other techniques.
Figure I. Fossils from the Herefordshire Lagerstätte. (A, B) The mollusc Kulindroplax (specimen length 38 mm; [74]); (A) photograph of a ground surface of the fossil; (B) digital reconstruction produced from serial grinding images. (C) Digital reconstruction of the fossil horseshoe crab Dibasterium (specimen length 23.2 mm excluding appendages; [75]).
Several techniques exist for nondestructively characterizing fossils in 3D (summarized in Table 1), but those that employ X-rays are by far the most common (Figure 1C, G–J). Indeed, the use of X-rays to study fossils has an even longer history than serial grinding; within months of their discovery in 1895, paleontologists had begun exploiting them to examine difficult-to-prepare material, such as the pyritized fossils of the Hunsrück Slate (Figure 1C) [15]. However, it was not until the late twentieth century that a tomographic approach was combined with X-ray techniques, enabling the use of X-ray computed tomography (CT) in paleontology (e.g., 16 and 17). This method works by obtaining a series of radiographs (projections) of a specimen at multiple angles as the specimen is penetrated by an X-ray beam. The resulting projections are then used to computationally generate a series of parallel slices perpendicular to the axis of rotation, which map X-ray attenuation through the specimen. The resulting dataset can be visualized and analyzed using a variety of software packages (summarized in Table 2). X-ray CT provides a nondestructive means of imaging 3D fossils, which is vital when studying rare or unique material.
| Technique | Data collected | Destructive? | Size of featureb | Resolutionc | Cost to buy/used | Portable? |
|---|---|---|---|---|---|---|
| Serial grinding, slicing and sawing | Optical images of exposed surfaces | Y | >1 mm | 10 μm | ££/£ | Y |
| FIB tomography | SEM images, chemical data | Y | 1 μm to 1 mm | 50 nm | ££££/£ | N |
| Micro-CT | Maps of X-ray attenuation | N | 1 mm to 250 mm | 1 μm | ££££/£ | N |
| Nano-CT | Maps of X-ray attenuation | N | 1 μm to 60 μm | 200 nm | ££££/£ | N |
| Synchrotron-based tomography | Maps of X-ray attenuation | N | 50 μm to 600 mm | 200 nm | X | N |
| Synchrotron-based phase contrast tomography | Maps of X-ray phase variations | N | 50 μm to 600 mm | 200 nm | X | N |
| Neutron tomography | Maps of neutron attenuation | N | 2 mm to 300 mm | 30 μm | X | N |
| Magnetic resonance imaging (MRI) | Distribution of light elements | N | <1 m | 10 μm | ££££/£ | N |
| Laser scanning | Surface color and morphology | N | >10 mm | 50 μm | ££/£ | Y |
| Photogrammetry | Surface color and morphology | N | Anye | N/Ae | £/£ | Y |
| Serial focus light microscopy | Optical images at successive planes | N | 100 μm to 10 mm | 200 nm | ££/£ | Y |
| Confocal laser scanning microscopy | Optical/fluorescence images at successive planes | N | 10 μm to 250 μm | 800 nm | £££/£ | N |
- a For a comprehensive comparison of available techniques, see [76].
- b The size range of the features that can be imaged with a technique; note, however, that for some techniques it may not be practical to obtain the highest resolution for the largest specimens.
- c The maximum resolution achievable on commonly used equipment; for techniques, such as serial grinding, the distance between slices is given, although the resolution within a single slice will be higher.
- d Cost of buying or manufacturing equipment/the cost per specimen of using third party equipment: £ = 10 s to 100 s of GB pounds; ££ = 100 s to 1000 s of GB pounds; £££ = 1000 s to 10 000 s of GB pounds; ££££ = 10 000 s to 100 000 s of GB pounds; X = time awarded competitively at no cost to the user.
- e Although there is no theoretical limit to the object size scaling in photogrammetry, for practical reasons the minimum size is in the millimeter range.
| Software package | Source | Example |
|---|---|---|
| CT data processing/image segmentation | ||
| Amira (www.amira.com) | Commercial | Donoghue et al.[23] |
| Avizo (www.vsg3d.com) | Commercial | Lautenschlager [77] |
| Drishti (http://anusf.anu.edu.au/Vizlab/drishti) | Freely available | Jones et al.[78] |
| Mimics (www.materialise.com/mimics) | Commercial | Domínguez Alonso et al.[39] |
| SPIERS (www.spiers-software.org) | Freely available | Sutton et al.[66] |
| VG Studio Max (www.volumegraphics.com) | Commercial | Butler et al.[79] |
| Visualization and animation | ||
| Autodesk Maya (http://usa.autodesk.com/maya) | Commercial | Molnar et al.[80] |
| Blender (www.blender.org) | Freely available | Stein [81] |
| 3D meshing/conversion of formats | ||
| GeoMagic Studio (www.geomagic.com) | Commercial | Arbour and Currie [82] |
| Altair Hypermesh (www.altairhyperworks.com) | Commercial | Lautenschlager et al.[83] |
| MeshLab (meshlab.sourceforge.net) | Freely available | White et al.[84] |
| ScanFE (http://www.simpleware.co.uk/) | Commercial | Young et al.[85] |
| Finite element analysis | ||
| Abaqus FEA (www.simulia.com) | Commercial | Lautenschlager et al.[83] |
| ANSYS (www.ansys.com) | Commercial | Witzel and Preuschoft [86] |
| COMSOL Multiphysics (http://www.uk.comsol.com/) | Commercial | Snively and Cox [87] |
| Strand7 (www.strand7.com) | Commercial | Walmsley et al.[88] |
| Computational fluid dynamics | ||
| ANSYS (www.ansys.com) | Commercial | Rigby and Tabor [53] |
| COMSOL Multiphysics (http://www.uk.comsol.com/) | Commercial | Caromel et al.[89] |
| SC/Tetra (http://www.cradle-cfd.com/) | Commercial | Shiino et al.[54] |
| SPHYSICS (https://wiki.manchester.ac.uk/sphysics/) | Freely available | Rahman and Falkingham [52] |
Early X-ray tomography studies mainly focused on digitally characterizing macroscopic fossils that had already been extracted from the host rock [16] (although see [17]). This was largely because distinguishing fossil remains from their host rock was problematic for the CT systems available at the time, which had originally been designed for noninvasive medical imaging and thus used low-energy polychromatic X-rays and short exposure times to minimize patient radiation dosage. Thus, they could not readily penetrate dense materials such as rocks. Furthermore, these medical CT scanners were restricted to relatively low-resolution imaging and thus could not visualize the fine anatomical details that were preserved in many important fossils, especially microscopic samples. In recent years, however, the utility of X-ray tomography for studying fossils has dramatically improved, with paleontologists taking advantage of the development of the high-energy and high-resolution variants micro-CT 18, 19 and 20 and, less frequently, nano-CT [21] to study a wide range of taxonomic groups and preservation types. These methods have made it possible to remove fossil specimens from their host rock entirely digitally (Box 2) and also to examine internal anatomy in great detail. Micro-CT has seen particularly broad usage because the technology is capable of achieving resolutions of a few microns or less and is applicable to a range of sizes and compositions; consequently, it is rapidly becoming a standard item of laboratory equipment at research institutions worldwide [22]. Nevertheless, the techniques typically have difficulty imaging chemically homogeneous samples that do not show substantial X-ray attenuation contrast, such as calcite fossils preserved in calcareous sediments (Box 1).
Box 2 – Digital restoration and reconstruction:
Fossils, by their very nature, are typically incomplete and/or distorted as a result of taphonomic processes, excavation, subsequent preparation, and handling. This can present a serious problem for the computational analysis of fossils, as many morphological and functional analyses require or rely on the original form, which might not be preserved. Thus, the outcome of such studies is closely correlated with the true-to-life correctness of the models used, and distorted or fragmentary specimens would inevitably yield incorrect results and lead to possibly erroneous conclusions [90]. Consequently, it is essential that the original pre-fossilization morphology is restored, before the shape or function can be rigorously analyzed. The restoration of fossil material, particularly of hominid crania, is standing practice in archeology and paleoanthropology. However, it is only recently that computational methods and digital models have replaced physical restoration techniques using photographs, drawings, and plaster models 91 and 92. Moreover, 3D restorations have only rarely been performed in paleontology 85, 93, 94 and 95.
Digital reconstruction and restoration techniques offer a variety of approaches to restore fossil morphology, ranging from the comparably simple removal of small breaks [96] and the replacement of missing elements or regions 80 and 97, to the realignment of disarticulated components [92] and the retrodeformation of whole fossils 82, 98 and 99. By employing a combination of these reconstructive steps, Lautenschlager [77] used CT scans of the original specimen (Figure IA, B) to restore the cranial skeleton of the Cretaceous therizinosaur dinosaur Erlikosaurus andrewsi ( Figure IC). The restored digital model was then subsequently used as a basis to reconstruct relevant soft tissue structures, such as the jaw adductor musculature (Figure ID) and a keratinous rhamphotheca covering the beak-like jaws. This combined reconstruction of hard and soft tissue ultimately permitted the investigation of feeding function in this herbivorous theropod dinosaur using FEA [77] (Figure IE, Figure 2).
Figure I. Individual steps in the digital restoration and reconstruction process exemplified by a model of the skull of Erlikosaurus andrewsi. (A) Original fossil (lower jaws omitted due to disarticulation). (B) Digital representation of the fossil. (C) Restored cranial anatomy. (D) Restored skull with reconstructed jaw adductor muscles. (E) Final finite element model based on (C) and (D). Skull length is 260 mm.
Digital reconstruction and restoration techniques provide powerful tools for the removal of taphonomic and preservational artifacts and hold an unprecedented potential to restore the antemortem state of a fossil. However, it is important to keep in mind that each reconstruction step increases the degree of interpretation introduced into the restored fossil, in particular if performed (often out of necessity) manually. Attempts to avoid this have been made by applying mathematically defined and repeatable techniques, in particular geometric morphometrics, and by automating the reconstruction methods. This makes it possible to create a distribution of reconstructions, for which the statistical significance, and thus the reliability, of a single reconstruction can be subjected to evaluation and discussion 91, 92 and 100.
Perhaps the most powerful of all X-ray tomographic methods is synchrotron-radiation tomography, where a cyclic particle accelerator is used to generate extremely bright X-rays. Synchrotron-radiation tomography has become increasingly popular in paleontology in recent years 23 and 24, partly because it is capable of rapidly scanning specimens at exceptionally high resolutions – surpassing those of micro-CT – but also due to the advantages provided by the monochromatic source. Current laboratory-based systems, including micro-CT, invariably employ polychromatic X-ray sources that emit a broad spectrum of X-ray energies, which can lead to scanning artifacts that can complicate the interpretation of fossil structures. By contrast, synchrotron sources emit only X-rays of a single energy (i.e., they are monochromatic), which enables better quality imaging with higher contrast and fewer artifacts compared to polychromatic sources. Furthermore, synchrotron-based phase-contrast imaging can improve the sensitivity with which minerals of similar densities can be resolved. This methodology exploits X-ray refraction at material boundaries, rather than conventional X-ray attenuation, in order to enhance the contrast between materials. In this way, difficult specimens that would not normally be amenable to X-ray tomography (i.e., those with low attenuation contrast) can be studied nondestructively in 3D at high resolution 25 and 26. At present, the availability of these cutting-edge techniques is limited by the small number of these international facilities. However, this seems set to change in the near future as the technology now exists for laboratory-based monochromatic sources [27] and phase-contrast imaging 28 and 29. Ultimately, these developments should mean that imaging only previously possible at synchrotron facilities will become routine in paleontology laboratories.
For some fossils, the resolutions that can be attained using X-ray tomography, even with a synchrotron, are insufficient to fully resolve the preserved details. In such cases, an alternative method can be applied: focused ion beam (FIB) tomography, which is capable of the highest resolution of any modern tomographic technique. FIB tomography entails in situ sequential milling and imaging of very small regions of interest; to date, it has been used only rarely in paleontology to characterize microscopic surfaces of very small fossils in 3D [30]. The method works in a similar way to traditional serial grinding, with successive faces physically exposed by a highly focused ion beam. The exposed faces are then imaged, frequently using a scanning electron microscope (SEM) coupled to the FIB instrument, and the resulting slice images are digitally aligned and reconstructed to produce a 3D model. The resolution that can be achieved is astonishing; the use of controlled ion milling means that slices can be less than 50 nm apart, and the spatial resolution within a single SEM slice provides nano-scale morphological information 30 and 31. Thus, although destructive and time-consuming, FIB tomography has enormous potential for the study of tiny microfossils and very fine features (e.g., microstructure and histology) of larger fossils, resolving anatomical details that could not be studied with any other 3D imaging method.
A handful of other tomographic methods have seen occasional use in paleontology. Neutron tomography is similar to X-ray CT, but relies on the differential absorption of neutrons (instead of X-rays) to image the interior of a specimen. Because neutrons are strongly attenuated by certain light elements (e.g., hydrogen) and readily penetrate many heavy elements (e.g., lead), this technique is suitable for large fossils in dense, metal-rich rocks and organically preserved specimens; however, the resolutions achieved are inferior to X-ray CT and, moreover, neutron bombardment can induce potentially hazardous levels of radioactivity in samples [32]. Magnetic resonance imaging (MRI) uses a strong magnetic field to map the distribution of certain elements (usually hydrogen) in a sample. It can therefore be used to image fossils with high hydrogen content (i.e., those with residual water, such as mummified specimens), but typically requires long scan times and generates relatively low-resolution images; it is better suited to imaging soft tissues in biological specimens [33]. Finally, optical tomography works by illuminating a light-transmitting sample in order to obtain images of successive planes through it. This can be achieved through serial focusing, where a conventional light microscope or a confocal laser-scanning microscope is used for imaging [34]. The approach is amenable to small specimens in a translucent matrix (e.g., fossils in chert or amber), but is not effective for opaque samples. All three of these methods are nondestructive, but are restricted to specific preservation types and/or require access to specialist equipment/facilities. These restrictions will most probably limit their uptake in paleontology.
In many cases, access to the interior of the fossil is not a requirement to paleontological study, for example, if only external surface features are of interest. Here, alternative nondestructive 3D imaging approaches can be employed such as surface scanning techniques that collect spatial and geometric data from a 3D surface at distance. These approaches have the advantage that the equipment is often cheap, portable, and easy to use (Table 1), which makes surface scanning suitable for characterizing fossils in field conditions (e.g., trace fossils; [35]), as well as for analyzing large museum specimens that cannot be moved (e.g., complete articulated vertebrate skeletons; 36 and 37) and thus are not suitable for study with any tomographic technique. Laser scanning is the most common surface-based method currently employed in paleontology and uses laser light to actively image the surface of interest. Photogrammetry passively reconstructs the geometric properties of an object from a series of overlapping photographs (or SEM images) taken at different orientations, providing information about the shape and color of the sample. Given that photogrammetry can, in principle, even be performed using a smartphone at resolutions comparable to standard laser scanning, this highly accessible technique has the potential for widespread adoption [37].
Digitally reconstructing paleobiology
Almost all the traditional problems associated with recovering fossil data from rocks can be overcome with modern 3D imaging. By making use of the full range of 3D imaging techniques now available to paleontologists, it is possible to obtain astonishingly high-resolution characterizations of fossils that can even exceed the limits of preservation. As a result, a broad range of previously intractable paleobiological questions can now be addressed. Tomographic techniques can be used to characterize the internal anatomy of three-dimensionally preserved fossils at nanometer and micrometer scale resolutions 22, 23 and 30, allowing the study of structures that would previously have been impossible to visualize and providing additional characters for comparisons with modern species and phylogenetic analyses [38]. In some cases, characterizing fossils in 3D enables inferences about their behavior to be made. For example, virtual endocasts of vertebrate braincases have been used to infer sensory and locomotory capabilities in extinct taxa 39, 40, 41 and 42.
These approaches can also be used to study the development of fossil organisms. In many taxa, from protists to problematic invertebrates and even vertebrates, the pattern of development is routinely preserved by the growth lines within the mineralized skeleton of the adult. CT can, therefore, be used to reconstruct the development of these organisms, by extracting the juvenile morphologies preserved within the skeletons of their adult selves 43, 44 and 45.
Furthermore, 3D data are invaluable for understanding the processes of fossilization. Paleontologists attempting to interpret exceptionally preserved organisms are perpetually confounded by the fact that such fossils have invariably been subjected to decay and are often overprinted by later phases of geological mineralization unrelated to the biology of the organism. Thus, it can be devilishly difficult to discriminate biology from geology, misleading researchers to overinterpret the extent of the biology preserved [46]. Fortunately, 3D imaging can help to tease out the original biological structure from these geological artifacts 31 and 47. Tomographic approaches can even be used to better understand the probable impact of decay on modern organisms [48], as well as to establish chemical or textural criteria that can distinguish between these phases of mineralization 47 and 49.
Finally, 3D imaging of fossils can provide the basis for digitally reconstructing fossil organisms with greater objectivity than was previously possible from the incomplete data that paleontologists are usually obliged to work with. Digital reconstruction, which involves digital restoration and retrodeformation of skeletal data and more objective reconstruction of soft tissue anatomy, has overhauled and revitalized studies of comparative musculoskeletal anatomy (Box 2).
Functional analysis of fossil organisms
Paleontologists have always had a fascination with how ancient organisms fed and moved, and what particular aspects of their anatomy were used for. Until recently, however, it was often difficult to study the function of extinct organisms in a way that allowed specific hypotheses to be tested. As a result, endeavors in this field have frequently been renounced as unscientific just-so stories that are no more rigorous than Kipling’s explanation for how the elephant acquired its trunk. However, tomographic approaches have provided a basis for formulating and exacting hitherto intractable tests, enhancing scientific rigor in the functional analysis of fossil organisms [50].
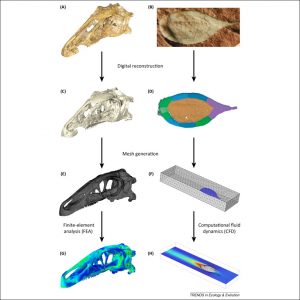
Functional analysis through computer modeling has developed in-hand with tomographic methods, not least because these analytical techniques depend on the availability of accurate 3D digital reconstructions. One such method is finite element analysis (FEA), an engineering approach that has been used in paleontology to help understand feeding and locomotion in fossil taxa. This approach can reconstruct stress, strain, and deformation in digital structures, including models of fossils [51]. This is achieved by converting the digital model into a finite element mesh, which divides the geometry of the object into numerous simple and discrete elements that are assigned life-like physical properties. When the virtual model is constrained and stressed in a manner compatible with known or hypothetical biomechanical performance, implied stress and strain can be computationally calculated, and validated using data not included in the model (e.g., detailed histology, microwear, in vivo or ex vivo experimentally recorded strain) ( [51]; Figure 2). Computational fluid dynamics (CFD) is a technique adopted from engineering that can be used to test various hypotheses relating to the performance of extinct organisms in aerial or aquatic environments 52, 53 and 54 (Figure 2). The quantification of morphology also enables the quantification of the motion that extinct organisms would have been capable of; for example, by studying the range of motion in joints [55], or by using multibody dynamics analysis (MDA), which models the motion and dynamic behavior of interconnected bodies such as muscles and bones 56 and 57.
Figure 2: Summary of the main steps involved in functional analysis through computer modeling. (A, C, E, G) Erlikosaurus andrewsi (specimen length 260 mm; [77]). (B, D, F, H) Protocinctus mansillaensis (specimen length 23 mm; [52]). (A, B) Photographs of original fossil specimens. (C, D) Digital reconstructions of fossils based on X-ray computed tomography. (E, F) Finite element meshes generated from digital reconstructions. (G) Finite element analysis of bite force performance. (H) Computational fluid dynamics simulation of hydrodynamic performance.
It is vital that the models used in all types of functional analyses are validated to assess their match to empirical data. This allows researchers to quantify how far estimated values may deviate from measured data before the model is applied to extinct organisms [58]. The data used to validate the model are usually derived from measurements taken from related living taxa [59]. However, another possible approach is to create a physical model of the specimen, which can now be easily produced from the digital dataset using 3D printing technology. The physical model can be loaded (for validating computational stress/strain analyses) or placed in a flume tank or wind tunnel (for validating models of fluid flow [53]), and the results compared to those of the digital analysis. Experimental loading of printed models has been applied to both real and hypothetical morphologies [60], and in the future it will probably be possible to print models with more life-like material properties. Sensitivity analysis is also important to determine the relation between the results and the input parameters, and to identify which of the parameters have the greatest influence on the output (and are therefore most important to quantify precisely) [58]. This is typically achieved by varying one input parameter at a time and evaluating the change in output. This type of careful use of techniques such as FEA, CFD, and MDA has enabled the replacement of studies rooted in anecdote, analogy, or scenarios, leading to scientific approaches where the relative ease of manipulation of digital data permits functional analysis within a hypothesis testing framework.
A virtual future
Digital datasets have been touted as a panacea for the problems of limited access to fossil specimens. In principle, they can be shared online to make them available to the entire community, providing paleontology with the openness enjoyed by other biological sciences. In reality, however, such global dissemination has rarely been realized 61 and 62. One reason for this is the unwillingness of some researchers to share their datasets until they have completed additional analyses. There would presumably be unanimous agreement that researchers must share enough data that other workers can verify the findings of a paper [63]. In the case of a paper describing new anatomical features based on tomographic data, this could be achieved by providing a down-sampled movie that shows each slice of a CT dataset in succession. There is less consensus regarding whether the data required to reproduce the study, including full-resolution slice data and segmentation files, should be released and, if so, how and when this should be done. Although some have argued for an embargo period to allow researchers to exploit the full potential of their data before sharing it [64], it is our contention (along with others [65]) that making these available at the time of publication should be mandatory. This should include both the primary tomographic data (to allow validation of the model) and any digital models (to enable repeatability of other analyses), presented in a widely used format [66]. However, the plethora of different (and often proprietary) file formats makes this more difficult to achieve in practice, and a standard file format has not yet been agreed upon. This will probably be dictated by the requirement for broad usage. A format such as STL (stereolithography), which describes a triangulated surface within a 3D Cartesian coordinate system and can be imported into a number of packages, including those associated with 3D printing, seems appropriate. Indeed, with the dramatic fall in the cost of 3D printers, such as the MakerBot models (New York, NY, USA), we now commonly receive requests from educators for the STL files associated with our published digital models so that they can print them for use in teaching. Although sharing the final digital models provides a considerable degree of transparency, for full traceability and reproducibility, further data, such as segmented labels and model parameters, should be included.
However, even when researchers are keen to share their data online, this is not always possible, forcing researchers (ourselves included) to make data available only by request. Firstly, there are legal issues regarding the ownership of the scan data. Before granting permission to carry out a scan, museums often require that researchers sign documents accepting that the data (including 3D models derived from them) remain the intellectual property of the institution [61], yet few museums have the infrastructure in place to store and disseminate such data. It is inevitable that such policies will have to be revisited as tensions emerge over publishing; museums, publishers, or third-party data repositories are going to have to invest in the technology and the policies for the open-access distribution of such data. Establishing and maintaining an online repository of tomographic data that is stable and secure in perpetuity will not be cheap because datasets can be very large. One recent study [67] was directly based on 140 GB of synchrotron-derived data – inevitably this will soon appear minimalistic. Although some online repositories for tomographic data already exist (e.g., www.paleo.esrf.eu, www.digimorph.com, www.datadryad.org), ideally all such data would be held in an open-access central repository. Analogous repositories, such as GenBank, have had to confront the increasing cost of data storage, both in terms of hardware and maintenance; it no longer accepts sequencing trace archives because it is now cheaper to resequence than store the data. It is very possible that, as CT hardware becomes more commonplace, this might be a preferred solution. Although we advocate the establishment of a central data repository, we suggest that it could make use of much cheaper, more stable, and greener technology in the form of automated tape archives. After all, it is our experience that individual high-resolution digital datasets are rarely reused. This may change with the potential discovery of instances of scientific fraud in the translation of tomographic datasets to virtual models, but it is unlikely that a single dataset is going to be required recurrently by concurrent users. Hence, demand for real-time access that can only be facilitated by disk or chip storage, which is both expensive to purchase and maintain, should be low. It is vital that these challenges are met if the promise of global sharing of digital fossil specimens among paleontologists is to be realized. There would also be wider benefits as digital models are a valuable tool in public engagement with science and teaching [68] because they allow rare fossils, including the very small and the very large and even those still embedded in rock [69], to be viewed in a dynamic and interactive way as well as to be printed out as physical models.
Most of the techniques we have discussed are currently being used to assess anatomy and functional morphology in individual taxa or in simplified digital datasets. However, as scanning becomes more commonplace, and as access to data improves and digital data-sharing grows, larger-scale multi-taxon studies will become increasingly feasible 70 and 71. The potential for assembling large quantities of comparative data means that the opportunity to assess broad-scale hypotheses pertaining to character evolution and the evolution of function and performance is now within reach. Future work, therefore, hinges on the assembly and maintenance of comparative databases.
In the future, the falling costs and increasing availability of CT scanners will mean that more paleontologists will have access to these techniques so that they can be used to address a wider range of questions for a variety of taxonomic groups. Future analyses are likely to become increasingly sophisticated, for example, allowing simultaneous physical, chemical, and textural analyses, allowing all these features to be studied and compared in three dimensions. Improvements in scanning and data processing techniques are likely to make it possible to obtain 3D characterizations of flattened fossils, such as those from the Hunsrück Slate, which were the subject of the first X-ray studies in paleontology [15]. The resolution that can be achieved is continuing to improve [72] and the time required for scanning is expected to keep decreasing. These advances will allow much more of evolutionary history to be read from the fossil record, and with ever-greater fidelity.
As a result of such ongoing advances, the characterization and analysis of fossil remains is now much more objective and reproducible than ever before. Some may argue that, given what came before, such advance was inevitable. However, the foremost limitations on reading the fossil record now lie principally, and somewhat ironically, with the poor state of knowledge of the anatomy of the living biota.
References
-
- 1
- E. Jarvik
- On the Dermal Bones. Sensory Canals and Pitlines of the Skull in Eusthenopteron foordi Whiteaves, with some Remarks on E. säve-söderberghi Jarvik
- Almqvist and Wiksell (1944)
- 2
- E. Jarvik
- On the Visceral Skeleton in Eusthenopteron with a Discussion of the Parasphenoid and Palatoquadrate in Fishes
- Almqvist and Wiksell (1954)
- 3
- P. Leiggi, P. May
- Vertebrate Palaeontological Techniques, Vol. 1, Cambridge University Press (1994)
- 4
- L. Jeppsson, et al.
- The optimal acetate buffered acetic acid technique for extracting phosphatic fossils
- J. Paleontol., 73 (1999), pp. 964–972
- 5
- J.A. Long, et al.
- Devonian arthrodire embryos and the origin of internal fertilization in vertebrates
- Nature, 457 (2009), pp. 1124–1127
- 6
- W.J. Sollas
- A method for the investigation of fossils by serial sections
- Philos. Trans. R. Soc. Lond. B: Biol. Sci., 196 (1904), pp. 257–263
- 7
- E.A. Stensiö
- The Downtonian and Devonian vertebrates of Spitsbergen. Part I. Family Cephalaspidae
- Skrifter Svalbard Nordishavet, 12 (1927), pp. 1–391
- 8
- H.M. Muir-Wood
- On the internal structure of some Mesozoic Brachiopoda
- Philos. Trans. R. Soc. Lond. B: Biol. Sci., 223 (1934), pp. 511–567
- 9
- R.P.S. Jefferies, D.N. Lewis
- English Silurian fossil Placocystites forbesianus and ancestry of vertebrates
- Philos. Trans. R. Soc. Lond. B: Biol. Sci., 282 (1978), pp. 205–323
- 10
- P. Janvier
- The brachial articulation and pectoral fin in antiarchs (Placodermi)
- Bull. Mus. Natl. d’Hist. Nat. 4e Sér., 17 (1995), pp. 143–162
- 11
- D.M. Kermack
- True serial-sectioning of fossil material
- Biol. J. Linn. Soc., 2 (1970), pp. 47–53
- 12
- J. Walton
- A method of preparing sections of fossil plants contained in coal balls or in other types of petrifaction
- Nature, 122 (1928), p. 571
- 13
- J. Galtier, T. Phillips
- The acetate peel technique
- T.P. Jones, N.P. Rowe (Eds.), Fossil Plants and Spores, The Geological Society (1999), pp. 67–70
- 14
- C. Poplin, A. de Ricqlès
- Technique for serial sectioning for the study of undecalcified fossils
- Curator, 13 (1970), pp. 7–20
- 15
- W. Branco
- Die Anwendung der Röntgenstrahlen in der Paläontologie. Abhandlungen der Königlich Preussischen Akademie der Wissenschaften
- Verlag der Königlichen Akademie der Wissenschaften (1906)
- 16
- J. Wind
- Computerized X-ray tomography of fossil hominid skulls
- Am. J. Phys. Anthropol., 63 (1984), pp. 265–282
- 17
- G.C. Conroy, M.W. Vannier
- Noninvasive three-dimensional computer imaging of matrix-filled fossil skulls by high-resolution computed-tomography
- Science, 226 (1984), pp. 456–458
- 18
- T. Rowe
- Coevolution of the mammalian middle ear and neocortex
- Science, 273 (1996), pp. 651–654
- 19
- S. Zamora, et al.
- Plated Cambrian bilaterians reveal the earliest stages of echinoderm evolution
- PLoS ONE, 7 (2012), p. e38296
- 20
- J.W. Hagadorn, et al.
- Cellular and subcellular structure of Neoproterozoic animal embryos
- Science, 314 (2006), pp. 291–294
-
- 21
- M.E. Collinson, et al.
- Did a single species of Eocene Azolla spread from the Arctic Basin to the southern North Sea?
- Rev. Palaeobot. Palynol., 159 (2010), pp. 152–165
- 22
- R.L. Abel, et al.
- A palaeobiologist’s guide to ‘virtual’ micro-CT preparation
- Palaeontol. Electron., 15 (2012), p. 6T
- 23
- P.C.J. Donoghue, et al.
- Synchrotron X-ray tomographic microscopy of fossil embryos
- Nature, 442 (2006), pp. 680–683
- 24
- P. Tafforeau, et al.
- Applications of X-ray synchrotron microtomography for non-destructive 3D studies of paleontological specimens
- Appl. Phys. A, 83 (2006), pp. 195–202
- 25
- E.M. Friis, et al.
- Phase-contrast X-ray microtomography links Cretaceous seeds with Gnetales and Bennettitales
- Nature, 450 (2007), pp. 549–552
- 26
- P. Tafforeau, T.A. Smith
- Nondestructive imaging of hominoid dental microstructure using phase contrast X-ray synchrotron microtomography
- J. Hum. Evol., 54 (2008), pp. 272–278
- 27
- K. Achterhold, et al.
- Monochromatic computed tomography with a compact laser-driven X-ray source
- Sci. Rep., 3 (2013), p. 1313
- 28
- C. Muehleman, et al.
- In-laboratory diffraction-enhanced X-ray imaging for articular cartilage
- Clin. Anat., 23 (2010), pp. 530–538
- 29
- S.W. Wilkins, et al.
- Phase-contrast imaging using polychromatic hard X-rays
- Nature, 384 (1996), pp. 335–338
- 30
- J.D. Schiffbauer, S. Xiao
- Paleobiological applications of focused ion beam electron microscopy (FIB-EM): an ultrastructural approach to the (micro)fossil record
- M. Laflamme (Ed.), et al., Quantifying the Evolution of Early Life: Numerical Approaches to the Evaluation of Fossils and Ancient Ecosystems, Springer (2011), pp. 321–354
- 31
- D. Wacey, et al.
- Taphonomy of very ancient microfossils from the ∼3400 Ma Strelley Pool Formation and ∼1900 Ma Gunflint Formation: new insights using a focused ion beam
- Precamb. Res., 220 (2012), pp. 234–250
- 32
- B. Winkler
- Applications of neutron radiography and neutron tomography
- Rev. Miner. Geochem., 63 (2006), pp. 459–471
- 33
- A. Ziegler, et al.
- Application of magnetic resonance imaging in zoology
- Zoomorphology, 130 (2011), pp. 227–254
- 34
- J.W. Schopf, A.B. Kudryavtsev
- Confocal laser scanning microscopy and Raman (and fluorescence) spectroscopic imagery of permineralized Cambrian and Neoproterozoic fossils
- M. Laflamme (Ed.), et al., Quantifying the Evolution of Early Life: Numerical Approaches to the Evaluation of Fossils and Ancient Ecosystems, Springer (2011), pp. 241–270
- 35
- K.T. Bates, et al.
- Three-dimensional modelling and analysis of dinosaur trackways
- Palaeontology, 51 (2008), pp. 999–1010
- 36
- K.T. Bates, et al.
- Estimating mass properties of dinosaurs using laser imaging and 3D computer modelling
- PLoS ONE, 4 (2009), p. e4532
- Full Text via CrossRef
- 37
- P.L. Falkingham
- Acquisition of high resolution three-dimensional models using free, open-source, photogrammetric software
- Palaeontol. Electron., 15 (2012), p. 1T
- 38
- J.A. Gauthier, et al.
- Assembling the squamate tree of life: perspectives from the phenotype and the fossil record
- Bull. Peabody Mus. Nat. Hist., 53 (2012), pp. 3–308
- 39
- P. Domínguez Alonso, et al.
- The avian nature of the brain and inner ear of Archaeopteryx
- Nature, 430 (2004), pp. 666–669
- 40
- S. Lautenschlager, et al.
- The endocranial anatomy of Therizinosauria and its implications for sensory and cognitive function
- PLoS ONE, 7 (2012), p. e52289
-
- 41
- L.M. Witmer, et al.
- Neuroanatomy of flying reptiles and implications for flight, posture and behaviour
- Nature, 425 (2003), pp. 950–953
- 42
- D.K. Zelenitsky, et al.
- Olfactory acuity in theropods: palaeobiological and evolutionary implications
- Proc. R. Soc. Lond. B: Biol. Sci., 276 (2009), pp. 667–673
- 43
- M. Rücklin, et al.
- Development of teeth and jaws in the earliest jawed vertebrates
- Nature, 491 (2012), pp. 748–751
- 44
- D.J.E. Murdock, et al.
- Ontogeny and microstructure of the enigmatic Cambrian tommotiid Sunnaginia Missarzhevsky, 1969
- Palaeontology, 55 (2012), pp. 661–676
- 45
- D.N. Schmidt, et al.
- Linking evolution and development: synchrotron radiation X-ray tomographic microscopy of planktic foraminifers
- Palaeontology, 56 (2012), pp. 741–749
- 46
- P.C.J. Donoghue, M.A. Purnell
- Distinguishing heat from light in debate over controversial fossils
- Bioessays, 31 (2009), pp. 178–189
- 47
- J.A. Cunningham, et al.
- Distinguishing geology from biology in the Ediacaran Doushantuo biota relaxes constraints on the timing of the origins of bilaterians
- Proc. R. Soc. Lond. B: Biol. Sci., 1737 (2012), pp. 2369–2376
- 48
- S.Y. Smith, et al.
- Virtual taphonomy using synchrotron tomographic microscopy reveals cryptic features and internal structure of modern and fossil plants
- Proc. Natl. Acad. Sci. U.S.A., 106 (2009), pp. 12013–12018
- 49
- S. Bengtson, et al.
- A merciful death for the “earliest bilaterian,” Vernanimalcula
- Evol. Dev., 14 (2012), pp. 421–427
- 50
- L. Hautier, et al.
- Skeletal development in sloths and the evolution of mammalian vertebral patterning
- Proc. Natl. Acad. Sci. U.S.A., 107 (2010), pp. 18903–18908
- 51
- E.J. Rayfield
- Finite element analysis and understanding the biomechanics and evolution of living and fossil organisms
- Annu. Rev. Earth Plant. Sci., 35 (2007), pp. 541–576
- 52
- I. Rahman, P. Falkingham
- Computational fluid dynamics: a powerful tool for elucidating function in fossil taxa
- Geol. Soc. Am. Progr. Abstr., 44 (2012), p. 372
- 53
- S. Rigby, G. Tabor
- The use of computational fluid dynamics in reconstructing the hydrodynamic properties of graptolites
- GFF, 128 (2006), pp. 189–194
- 54
- Y. Shiino, et al.
- Swimming capability of the remopleuridid trilobite Hypodicranotus striatus: hydrodynamic functions of the exoskeleton and the long, forked hypostome
- J. Theor. Biol., 300 (2012), pp. 29–38
- 55
- S.E. Pierce, et al.
- Three-dimensional limb joint mobility in the early tetrapod Ichthyostega
- Nature, 486 (2012), pp. 523–526
- 56
- P. O’Higgins, et al.
- Combining geometric morphometrics and functional simulation: an emerging toolkit for virtual functional analyses
- J. Anat., 218 (2011), pp. 3–15
- 57
- K.T. Bates, P.L. Falkingham
- Estimating maximum bite performance in Tyrannosaurus rex using multi-body dynamics
- Biol. Lett., 8 (2012), pp. 660–664
- 58
- J.R. Hutchinson
- On the inference of function from structure using biomechanical modelling and simulation of extinct organisms
- Biol. Lett., 8 (2012), pp. 115–118
- 59
- J.A. Bright, E.J. Rayfield
- Sensitivity and ex vivo validation of finite element models of the domestic pig cranium
- J. Anat., 219 (2011), pp. 456–471
- 60
- S.B. Crofts, A.P. Summers
- How to best smash a snail: the effect of tooth shape on crushing load
- J. R. Soc. Interface, 11 (2014), p. 20131053
-
- 61
- J.J. Hublin
- Free digital scans of human fossils
- Nature, 497 (2013), p. 183
- 62
- T. Rowe, L.R. Frank
- The disappearing third dimension
- Science, 331 (2011), pp. 712–714
- 63
- B. Hanson, et al.
- Making data maximally available
- Science, 331 (2011), p. 649
- 64
- S. Portugal, S. Pierce
- Who’s looking at your data?
- Sci. Careers (2014) http://dx.doi.org/10.1126/science.caredit.a1400052
- 65
- A. Ziegler, et al.
- Opportunities and challenges for digital morphology
- Biol. Direct, 5 (2010), p. 45
- 66
- M.D. Sutton, et al.
- SPIERS and VAXML; a software toolkit for tomographic visualisation and a format for virtual specimen interchange
- Palaeontol. Electron., 15 (2012), p. 5T
- 67
- T. Huldtgren, et al.
- Fossilized nuclei and germination structures identify Ediacaran ‘animal embryos’ as encysting protists
- Science, 334 (2011), pp. 1696–1699
- 68
- I.A. Rahman, et al.
- Virtual fossils: a new resource for science communication in paleontology
- Evol. Educ. Outreach, 5 (2012), pp. 635–641
- 69
- R. Schilling, et al.
- Reviving the dinosaur: virtual reconstruction and three-dimensional printing of a dinosaur vertebra
- Radiology, 270 (2014), pp. 864–871
- 70
- V. Allen, et al.
- Linking the evolution of body shape and locomotor biomechanics in bird-line archosaurs
- Nature, 497 (2013), pp. 104–107
- 71
- A.M. Balanoff, et al.
- Evolutionary origins of the avian brain
- Nature, 501 (2013), pp. 93–96
- 72
- M. Dierolf, et al.
- Ptychographic X-ray computed tomography at the nanoscale
- Nature, 467 (2010), pp. 436–439
- 73
- M.D. Sutton, et al.
- Methodologies for the visualization and reconstruction of three-dimensional fossils from the Silurian Herefordshire Lagerstätte
- Palaeontol. Electron., 4 (2001) art. 1
- 74
- M.D. Sutton, et al.
- A Silurian armoured aplacophoran and implications for molluscan phylogeny
- Nature, 490 (2012), pp. 94–97
- 75
- D.E.G. Briggs, et al.
- Silurian horseshoe crab illuminates the evolution of arthropod limbs
- Proc. Natl. Acad. Sci. U.S.A., 109 (2012), pp. 15702–15705
- 76
- M.D. Sutton, et al.
- Techniques for Virtual Palaeontology
- Wiley (2014)
- 77
- S. Lautenschlager
- Cranial myology and bite force performance of Erlikosaurus andrewsi: a novel approach for digital muscle reconstructions
- J. Anat., 222 (2013), pp. 260–272
- 78
- M.E.H. Jones, et al.
- Hard tissue anatomy of the cranial joints in Sphenodon (Rhynchocephalia): sutures, kinesis, and skull mechanics
- Palaeontol. Electron., 14 (2011), p. 17A
- 79
- R.J. Butler, et al.
- Reassessment of the evidence for postcranial skeletal pneumaticity in Triassic archosaurs, and the early evolution of the avian respiratory system
- PLoS ONE, 7 (2012), p. e34094
- 80
- J.L. Molnar, et al.
- Idealized landmark-based geometric reconstructions of poorly preserved fossil material: a case study of an early tetrapod vertebra
- Palaeontol. Electron., 15 (2012), pp. 1–18
-
- 81
- M. Stein
- A new arthropod from the Early Cambrian of North Greenland, with a ‘great appendage’-like antennula
- Zool. J. Linn. Soc., 158 (2010), pp. 477–500
- 82
- V.M. Arbour, P.J. Currie
- Analyzing taphonomic deformation of ankylosaur skulls using retrodeformation and finite element analysis
- PLoS ONE, 7 (2012), p. e39323
- 83
- S. Lautenschlager, et al.
- Edentulism, beaks and biomechanical innovations in the evolution of theropod dinosaurs
- Proc. Natl. Acad. Sci. U.S.A., 110 (2013), pp. 20657–20662
- 84
- M. White, et al.
- Morphological comparisons of metacarpal I for Australovenator wintonensis and Rapator ornitholestoides: implications for their taxonomic relationships
- Alcheringa, 37 (2013), pp. 1–7
- 85
- M.T. Young, et al.
- Cranial biomechanics of Diplodocus (Dinosauria, Sauropoda): testing hypotheses of feeding behaviour in an extinct megaherbivore
- Naturwissenschaften, 99 (2012), pp. 637–643
- 86
- U. Witzel, H. Preuschoft
- Finite-element model construction for the virtual synthesis of the skulls in vertebrates: case study of Diplodocus
- Anat. Rec. A, 283A (2005), pp. 391–401
- 87
- E. Snively, A. Cox
- Structural mechanics of pachycephalosaur crania permitted head-butting behavior
- Palaeontol. Electron., 11 (2008), p. 3a
- 88
- C.W. Walmsley, et al.
- Why the long face? The mechanics of mandibular symphysis proportions in crocodiles
- PLoS ONE, 8 (2013), p. e53873
- 89
- A.G.M. Caromel, et al.
- Hydrodynamic constraints on the evolution and ecology of planktic foraminifera
- Mar. Micropaleontol., 106 (2014), pp. 69–78
- 90
- C. Foth, O.W.M. Rauhut
- The good, the bad, and the ugly: the influence of skull reconstructions and intraspecific variability in studies of cranial morphometrics in theropods and basal saurischians
- PLoS ONE, 8 (2013), p. e72007
- 91
- C.P.E. Zollikofer, M.S. Ponce de León
- Virtual Reconstruction: A Primer in Computer-assisted Paleontology and Biomedicine
- Wiley-Interscience (2005)
- 92
- P. Gunz, et al.
- Principles for the virtual reconstruction of hominin crania
- J. Hum. Evol., 57 (2009), pp. 48–62
- 93
- S.D. Sampson, L.M. Witmer
- Craniofacial anatomy of Majungasaurus crenatissimus (Theropoda: Abelisauridae) from the Late Cretaceous of Madagascar
- J. Vertebr. Paleontol., 27 (2007), pp. 32–33
- 94
- P.C. Sereno, et al.
- Structural extremes in a Cretaceous dinosaur
- PLoS ONE, 2 (2007), p. e1230
- 95
- F.J. Degrange, et al.
- Mechanical analysis of feeding behavior in the extinct “terror bird” Andalgalornis steulleti (Gruiformes: Phorusrhacidae)
- PLoS ONE, 5 (2010), p. e11856
- 96
- S. Lautenschlager
- Paleontology 2.0 – A comprehensive protocol for the reconstruction of hard- and soft tissue structures in fossils
- Geol. Soc. Am. Progr. Abstr., 44 (2012), p. 372
- 97
- F.E. Grine, et al.
- Reconstruction of the late Pleistocene human skull from Hofmeyr, South Africa
- J. Hum. Evol., 59 (2010), pp. 1–15
- 98
- N. Ogihara, et al.
- Computerized restoration of nonhomogeneous deformation of a fossil cranium based on bilateral symmetry
- Am. J. Phys. Anthropol., 130 (2006), pp. 1–9
- 99
- A.A. Boyd, R. Motani
- Three-dimensional re-evaluation of the deformation removal technique based on “jigsaw puzzling”
- Palaeontol. Electron., 11 (2008), p. 7A
- 100
- C. Zollikofer
- A computational approach to paleoanthropology
- Evol. Anthropol., S1 (2002), pp. 64–67
- 101
- Z.K. Gai, et al.
- Fossil jawless fish from China foreshadows early jawed vertebrate anatomy
- Nature, 476 (2011), pp. 324–327