Wikimedia Commons
By (left-to-right) Dr. Thomas C. Scott-Phillips, Dr. Thomas E. Dickins, and Dr. Stuart A. West
Scott-Phillips: Senior Research Fellow in Cognitive and Evolutionary Anthropology, Durham University
Dickins: Professor of Evolutionary Psychology, University of East London
West: Professor of Evolutionary Biology, University of Oxford
Abstract
To properly understand behavior, we must obtain both ultimate and proximate explanations. Put briefly, ultimate explanations are concerned with why a behavior exists, and proximate explanations are concerned with how it works. These two types of explanation are complementary and the distinction is critical to evolutionary explanation. We are concerned that they have become conflated in some areas of the evolutionary literature on human behavior. This article brings attention to these issues. We focus on three specific areas: the evolution of cooperation, transmitted culture, and epigenetics. We do this to avoid confusion and wasted effort—dangers that are particularly acute in interdisciplinary research. Throughout this article, we suggest ways in which misunderstanding may be avoided in the future.
Animal Behavior: An Evolutionary Approach, Tenth Edition / John Alcock
The difference between proximate and ultimate explanations of behavior is central to evolutionary explanation (Mayr, 1963; Tinbergen, 1963). Ultimate explanations are concerned with the fitness consequences of a trait or behavior and whether it is (or is not) selected. In contrast, proximate explanations are concerned with the mechanisms that underpin the trait or behavior—that is, how it works. Put another way, ultimate explanations address evolutionary function (the “why” question), and proximate explanations address the way in which that functionality is achieved (the “how” question). Another way to think about this distinction is to say that proximate mechanisms are behavior generators, whereas ultimate functions explain why those behaviors are favored. Examples can be found in any introductory textbook to evolution and behavior (e.g., Alcock, 2009; Krebs & Davies, 1993). Here is one from a textbook on evolution and psychology (Nettle, 2009c; drawing on Zeifman, 2001). Human infants cry. The ultimate explanation of this is that it elicits care and defense from mothers and other caregivers. This is an ultimate explanation because it appeals to the fitness benefits of the trait: Infants that do not cry when in need of assistance are less likely to survive. Proximate explanations include both the external triggers of crying, for example physical separation from the caregiver, cold, or a lack of food, and also the internal mechanisms, such as the limbic system and the endogenous opioids involved in the cessation of crying. These are proximate explanations because they describe the immediate causal triggers of crying. Note that our answer to one of these questions does not commit us to any particular answer to the other. They are not opposite ends of a continuum, and we should not choose between them. On the contrary, they are distinct from one another and complementary. To completely understand behavior, we must obtain both.
The evolutionary literature on human behavior contains many good applications of this distinction. One example, from the earliest days of evolutionary psychology, isCosmides’s work (1989) on human reasoning, in which she made a conjecture about ultimate functionality (that the ability to reason about social exchange would be selected for in a social species like humans) and then postulated a particular proximate mechanism that would perform this task (a specialized cognitive module for social exchange). The specifics of this claim have since been much discussed, and it is possible that it is incorrect. As with any scientific paradigm, the hypotheses that flow from an evolutionary perspective will often turn out to be false. This is part of the routine progress of science (Kuhn, 1962). What we wish to bring attention to, however, is the clear distinction between Cosmides’s claims about ultimate function on the one hand and about proximate mechanism on the other.
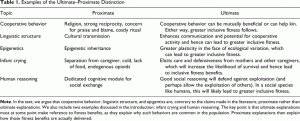
The distinction between ultimate and proximate explanations is central to the neo-Darwinian paradigm, yet we are concerned that the evolutionary literature on human behavior exhibits some confusion about it. In particular, (a) in the literature on the evolution of cooperation, proximate explanations are often offered as solutions to the ultimate problem of why an individual should do something that benefits another; (b) in the literature on cultural evolution, it is sometimes claimed that the process of cultural evolution offers an ultimate explanation, whereas we argue that it is better understood as a proximate mechanism; and (c) epigenetic phenomena are often said to challenge natural selection (an ultimate process) as a source of explanation, but again, these are better understood as proximate mechanisms. Table 1 compares the examples of infant crying and social reasoning with these three areas.
Table 1: Click Image to Enlarge
Our objective in this article is to bring attention to these issues. We do this not to simply point out errors, but because we are concerned that the confusions we highlight hinder scientific progress. As such, the potential for wasted effort is significant, particularly in interdisciplinary research, such as evolutionary psychology and human behavioral ecology, in which nuances in meaning and/or explanatory status can be lost. We therefore discuss possible underlying reasons for the confusions that we highlight and suggest several ways in which they may be avoided in the future. To provide a foundation for the subsequent discussion, the next section charts the historical development of evolutionary theory since Darwin, and uses that to illustrate how the proximate–ultimate distinction is a direct consequence of the theory of natural selection. The three sections that follow it then address the three topics mentioned above (the evolution of cooperation, cultural transmission, and epigenetics).
Historical Development of Darwinism, and the Ultimate-Proximate Distinction
Darwin’s (1859) key insight was that those heritable traits that are associated with greater reproductive success will, over time, tend to accumulate in a population, and that those associated with reduced reproductive success will disappear. This explains both how natural selection works (a process that will favor traits that increase reproductive success) and also what its consequences will be (organisms that appear to be designed to maximize fitness; Gardner, 2009). This distinction between the process and its consequences is at the heart of the ultimate–proximate distinction.Fisher (1930), Haldane (1932), and Wright (1931) synthesized Mendelian genetics with Darwin’s theory and, in doing so, showed that the process of natural selection works through changes in gene frequency (Provine, 2001).
The Extended Phenotype: The Long Reach of the Gene / Richard Dawkins
Fisher’s (1930) fundamental theorem of natural selection formalized the insight that natural selection will produce organisms that appear to be designed to maximize their fitness (i.e., they behave as maximizing agents). Within this framework, fitness is defined as the number of offspring an individual produces that survive to reproductive age (Dawkins, 1982; Grafen, 2007; Hamilton, 1964). Although this is an appropriate measure of fitness for many scenarios, it fails for social behaviors when interacting individuals are more (or less) likely to share genes than expected by chance, such as when interactions take place between relatives who share genes from a common ancestor. In this case, we must allow for the fact that a gene can also influence its transmission to the next generation indirectly by influencing the reproductive success of other individuals who carry that gene.
Hamilton’s contribution (1964, 1970, 1975) was to incorporate these indirect effects into the theory of natural selection. He showed that a gene’s increase in frequency is due not only to the direct fitness effects that it has on the focal individual, but also to the indirect fitness effects that it has on the fitness of the focal individual’s relatives. These indirect effects are famously captured in Hamilton’s rule, which states that a behavior or trait will be favored by selection when rb > c, where c is the fitness cost to the actor, b is the fitness benefit to the recipient, and r is their genetic relatedness. The coefficient of relatedness (r) is a statistical concept, describing the genetic similarity between two individuals relative to the average similarity of all individuals in the population (Grafen, 1985; Hamilton, 1970).1
The sum of direct and indirect fitness is called inclusive fitness. Consequently, inclusive fitness is the quantity that organisms should appear to maximize. Put another way, inclusive fitness is the maximand of natural selection (Grafen, 2006). Inclusive fitness is not a special case; it is instead our most general encapsulation of Darwinian fitness (Grafen, 2007; Grafen 2009). One related and often misunderstood term is kin selection. Coined by Maynard Smith (1964), kin selection is usually used to mean selection of the indirect component of inclusive fitness (West, Griffin, & Gardner, 2007). As such, kin selection is not an alternative to natural selection as a source of explanation. Rather, it is part of natural selection (Dawkins, 1979; Grafen, 2006; West et al., 2007). It should also be emphasized that methods such as multilevel (group) selection do not give different predictions—they are just alternate ways of analyzing the dynamics of natural selection, and they still lead to the maximization of inclusive fitness (Hamilton, 1975). Put simply, nothing else has been shown to be a maximand of natural selection.
Inclusive fitness, then, is the metric by which natural selection operates. If one variant of a trait contributes more to inclusive fitness than some other variant, then this explains why that variant that is common in the population. Such explanations of behavior are ultimate explanations, as they appeal to fitness consequences and explain why (or why not) that trait is common in the population. Put simply, if we wish to offer an ultimate explanation for the existence of some trait, we must make reference to how that trait contributes to inclusive fitness.
That does not mean that the ultimate question always has the same answer: Traits can contribute to inclusive fitness in a variety of ways. For example, infant crying contributes to inclusive fitness because it informs adults that the infant is in distress and hence elicits care from caregivers. Infant smiling, on the other hand, may contribute to inclusive fitness because it increases the infant’s bond with the adult, who may hence be drawn to supplying more care to the infant in the future. (This proposal is pure conjecture—it is there only to illustrate that there are different types of ultimate explanation.) Exactly how a given trait works (in terms of the various social, psychological, cognitive, chemical, and physical mechanisms that are involved in its operation) is a separate and complementary question. Specifically: it is a proximate question.
Note that the ultimate–proximate distinction also applies to comparative work (Harvey & Pagel, 1991). For example, we might ask why humans cooperate in large groups but chimpanzees do not. One way to approach this question would be to ask about the effects of cooperative behavior on the inclusive fitness of humans and chimpanzees respectively. One possible answer to this might be that humans interact with one another frequently because they live in large social groups, and thus it is adaptive for the recipient of the cooperative act to reciprocate in the future rather than defect and suffer the consequences of a bad reputation and possible social ostracism. Consequently, humans can expect cooperative behavior to be reciprocated, and such behavior may result in an increase in inclusive fitness. Chimpanzees, on the other hand, live in fission–fusion societies, in which the likelihood of future interaction and reciprocation is reduced. Consequently, for chimpanzees cooperative behavior may often not lead to fitness benefits (at least, not in way as hypothesized here). Whether this story is right or not is not the point (in fact, it is almost certainly too simple, at best). The point is rather that this sort of answer is of an ultimate answer. A different way to approach the question of how humans differ from chimpanzees with respect to cooperation in large groups would be to ask about the proximate mechanisms involved. One possible answer would involve a description of the cognitive processes involved in cooperative behavior, which would almost certainly be different in humans and in chimpanzees. In sum, comparisons (whether across species or of some other sort) do not dilute the proximate–ultimate distinction. On the contrary, different comparisons can provide different sorts of explanations.
Unfortunately, there are many cases in the evolutionary literature on humans in which proximate mechanisms are used to address issues of ultimate explanation. The next three sections document some of these.
The Evolution of Cooperation
Proximate Phenomena Used to Address Ultimate Questions
One area in which inclusive fitness theory has been very fruitfully applied is in the evolution of cooperation, in which it has been used to explain cooperation in an extremely wide range of organisms. The central theoretical question for the evolution of cooperation is why an individual would perform a behavior that is beneficial to another (Hamilton, 1964). This is an ultimate level problem—it asks why such behaviors should exist at all. It thus demands an ultimate level solution. However, much of the human literature has suggested various proximate mechanisms as solutions to this ultimate problem. These include religion (Johnson, 2005; Johnson & Krueger, 2004); a concern for praise and blame (Ellingsen & Johannesson, 2007); costly ritual (Power, 2009); and psychological predispositions to reward cooperators and punish noncooperators, otherwise known as strong reciprocity (de Quervain et al., 2004; Fehr & Fischbacher, 2003; Fehr & Rockenbach, 2004).
All of these describe proximate phenomena: They explain how cooperation might work. For example, one suggestion (Johnson, 2005; Johnson & Krueger, 2004) is that the fear of supernatural punishment motivates individuals to behave cooperatively. This fear and the subsequent motivation are psychological phenomena—they explain (if the hypothesis is correct) how humans come to behave cooperatively. They do not, however, explain why such behavior is present in the population. It may, for example, be the case that individuals who behave cooperatively because of the fear of supernatural punishment have lower inclusive fitness than those who do not fear punishment and hence do not behave cooperatively—in which case such fear would be selected against. To explain why such fear exists in the first place, we must make reference to inclusive fitness effects.
Further examples are numerous. Here is one: “Human altruism is considered an evolutionary puzzle… here, we investigate the hypothesis that altruism is caused by feelings of shame and pride and that these feelings are accentuated by others’ verbal evaluation” (Ellingsen & Johannesson, 2007, p. 100). Similarly, “Why [from an evolutionary perspective] are unrelated members of human social groups who only infrequently interact so ready to cooperate with each other?… The only medium for securing such cooperation… is costly ritual” (Power, 2009, p. 257). In both of these cases, the question is posed from an evolutionary (i.e., ultimate) perspective, but a proximate mechanism is then offered as an answer. Another example is the literature on strong reciprocity. Strong reciprocity is defined as a combination of “a predisposition to reward others for cooperative, norm-abiding behaviors… [and] a propensity to impose sanctions on others for norm violations” (Fehr & Fischbacher, 2003, p. 785)—this defines it as a proximate mechanism. Yet it is offered as a solution to the ultimate problem of why humans cooperate (West, El Mouden, & Gardner, in press). The neurological work on strong reciprocity makes the same error (West et al., in press). It is not that the above-cited studies are worthless; on the contrary, they can help explain how cooperative behavior can lead to direct and indirect fitness benefits. Our point is only that they describe how cooperation might work and not why it exists. Similar issues occur in the literature on the evolution of language (Scott-Phillips, 2007).
What Sort of Explanations Are Being Sought?
Why are these misunderstandings relatively common? We suggest three possible reasons. The first is that proximate explanations are often (in fact, usually) the desideratum for research in the human behavioral sciences, and so the question of ultimate functionality is often not raised. A second (and related) reason may be that some terms have two different meanings: one at the ultimate level and another at the proximate level. For example, cooperation, altruism, spite, and associated terms are defined in the biological literature in terms of their net effects on direct fitness (Hamilton, 1964, 1970; West et al., 2007; see Table 2 ). As such, these theoretical constructs exist at the ultimate level of explanation. However, the same terms in the psychological literature are often used to refer to mental states, which are proximate mechanisms. These different frames of reference (direct fitness effects on the one hand, mental states on the other) are to be expected, given the different domains of enquiry. However, there is an associated danger that such terms may be misunderstood and/or misused when they are transferred between disciplines. It is not that certain uses of terms are better than others, but whichever is being used must be made clear if confusion is to be avoided. Moreover, the two must not be mixed.
Table 2: Click Image to Enlarge
To take a precise example, altruism is usually defined within psychology as a mental disposition to behave favorably to others. Yet the behaviors that result from this disposition will often produce direct fitness benefits for the actor. In particular, this will be the case if the favorable behavior is reciprocated in the future. As such, although such behaviors would be called altruistic from a psychological perspective, they will be mutually beneficial (i.e., beneficial to both actor and recipient in terms of contribution to direct fitness; see Table 1) from an evolutionary perspective. Consequently, when motivations to help others and associated dispositions are the target of evolutionary research (e.g., de Waal, 2006; Tomasello, 2009; Warneken & Tomasello, 2006), it is important to make clear the sense in which terms like altruism are being used (see alsoSober, 1988).
Primates and Philosophers: How Morality Evolved / Frans de Waal
This problem is sometimes exacerbated by the (often necessary) use of proxy measures for fitness—for example, financial payoffs or attractiveness judgments. There are often good methodological reasons why researchers use these measures—for one thing, it is usually impossible to conduct direct, controlled experimental research on how inclusive fitness is affected by the trait in question. However, the use of proxy measures carries the risk that the costs and benefits observed at this proximate level of analysis may not translate into costs and benefits at the ultimate level of analysis (i.e., in terms of inclusive fitness effects). For example, in particular economic games, cooperative individuals may receive greater payoffs than uncooperative individuals, but these payoffs may not necessarily translate into inclusive fitness effects. Yet this proviso is rarely made explicit.
The Use of Intentional Language in Evolutionary Explanation
A third possible explanation for proximate–ultimate confusion in the human literature may be the intentional language employed to describe behavior. Specifically, evolutionary biologists, behavioral ecologists, and other students of animal behavior often use intentional language to describe behavior, but they do not mean to suggest that members of the species in question have intentions in the way we would recognize them. Phrases like “offspring are selected to demand more food than the parent wants to give” are standard, but they are not intended to suggest that organisms (of whatever species) entertain notions of desire (“wants”) or imperatives (“demand”). Intentional language is employed as shorthand. The same point could in principle be expressed more neutrally: “During the course of evolution selection acting on genetic differences in the begging behaviour of offspring will have favoured an increase in the intensity of begging, and this will have been favoured to the extent that the level of begging by any individual offspring exceeds the optimum level for the parent” (example taken from Krebs & Davies, 1993, p. 3). This is clearly cumbersome, but it does make the functional nature of the claim more transparent.
This use of intentional language is justified by the fact that natural selection leads to organisms that behave as if they were intentional agents that seek to maximize inclusive fitness (Dawkins, 1982; Grafen, 1999).2 However, we should keep in mind that the justification comes from an ultimate level of analysis—organisms often appear to behave as if they have intentions or purpose at the individual level because the dynamics of natural selection lead to individual organisms that maximize fitness (see the previous section). The problem is that this sort of language invites misunderstanding when applied to humans, because humans have real, proximate intentions that are described using the same language as these ultimate explanations (Brunnander, 2008). For instance, the meaning of “females want to mate with the highest status male they can find” will depend on whether the person making that claim is speaking as an evolutionary biologist or a behavioral psychologist: The former is referring to behavioral strategies that maximize fitness, and the latter is referring to psychological motivations. Yet these two levels of analysis may not always correspond with one another.
The dangers discussed here are in fact doubly problematic. Not only do humans have intentions in the psychological sense, as discussed earlier, but there is also the potential issue that many readers of evolutionary claims about human behavior will be human behavioral scientists who, by virtue of their training, may not be aware that intentional language is often used in evolutionary discussions as anthropomorphic shorthand for ultimate explanations. Definitions grounded in evolutionary theory have the advantage of explicitly stating how human behavior relates to the behavior of all other organisms, but the disadvantage of not readily linking to the descriptive and/or explanatory frameworks of psychology and other disciplines concerned with human behavior. Either way, there is significant potential for misunderstanding in such interdisciplinary enterprises. So as to avoid confusion in the future, recent discussions in the animal communication literature (Rendall, Owren, & Ryan, 2009; Soltis, 2009) make a plea that functional claims should be made in more explicitly functional terms and that intentional language should be avoided. We do not go so far in our recommendation, but we do suggest that when humans are studied from an evolutionary perspective, researchers should take particular care in their use of intentional language and make explicit the sense in which it is used.
Culture and the Ultimate-Proximate Distinction
Cultural Transmission as a Proximate Mechanism
Culture and the Evolutionary Process / Robert Boyd and Peter J. Richerson
We turn now to the question of how the ultimate–proximate distinction applies to processes of cultural transmission and social learning.3 Following the established usage in the evolutionary literature (see e.g., Boyd & Richerson, 1985; Richerson & Boyd, 2005), we define transmitted culture as those behaviors or traditions that individuals acquire through teaching, imitation, and other forms of social transmission (see Nettle, 2009a, for a discussion of how this relates to other, nonevolutionary notions of culture). This transmitted culture is ubiquitous in human life. Textile design, fashion, social taboos, linguistic structure … the list of possible examples is almost endless. Humans are not unique in this regard, but they are certainly the most voracious users of transmitted culture—so much so that the effects of transmitted culture are crucial to a proper understanding of human behavior.
How should cultural transmission be understood with respect to the ultimate–proximate distinction? In short, we believe that processes of cultural transmission are best understood as proximate mechanisms (see also Nettle, 2009b). At least some of the scholars who have developed models of cultural evolution and its interaction with biological evolution agree: “these theories [of how cultural evolution interacts with biology] model the proximate mechanisms that produce adaptations” (Henrich & McElreath, 2003, p. 124). This view is not, however, universal. For example, while discussing how social psychology and cultural evolution can inform one another,Mesoudi (2009) wrote: “Traditional social psychological explanations tend to explain behavior in terms of proximate situational cues or internal motivations, while evolutionary theories [including cultural evolutionary theories] provide ultimate explanations in terms of when and why a behavior may have emerged in a species’ evolutionary past” (p. 933). A more indirect illustration of the view we wish to take issue with comes from discussion of the role of transmitted culture in the evolution of linguistic structure: “The challenge…is to determine for each feature of language that we wish to explain whether natural selection or iterated learning [a particular form of cultural transmission, in which individuals acquire behavior having observed similar behaviors in other individuals who acquired it in the same way; see next subsection, below] is the right explanatory mechanism” (Kirby, 2007, p. 678). As natural selection is uncontroversially an ultimate source of explanation, this attempt to contrast it with cultural transmission is in effect a claim that cultural transmission is also an ultimate source of explanation. We discuss this example of linguistic structure in the next section.
The reason why cultural transmission should be understood as a proximate mechanism is that it describes one of the causal triggers of the expression of some behavior. If the expression of some culturally transmitted variant (for example, a particular linguistic feature or fashion trend) is in part explained by the fact that it has been culturally transmitted, then this transmission process is one of the causal triggers of the expression of that variant—and as such, it is one of the many proximate mechanisms that contribute to a complete understanding of the variant under discussion. An ultimate explanation of the expression of said cultural variant would refer to how such behavior contributes to inclusive fitness (i.e., to the fitness benefits or costs that the expression of that variant confers upon the individual that has used it).
Animal Signals / John Maynard Smith and David Harper
One of the reviewers of this article suggested that culture cannot be only a proximate mechanism because the emergence of human culture changed the selection pressures that acted on humans (for example, the advent of dairy farming led to an increase in the frequency of genes for lactose tolerance; Holden & Mace, 1997; Tishkoff et al., 2007). However, this fact is irrelevant to our argument. The emergence of particular proximate mechanisms often changes the selection pressures that act upon a species. Consider, for instance, the well-documented process of ritualization, by which cues evolve into signals. One specific example is the use of urine to mark territorial boundaries (see Maynard Smith & Harper, 2003). Initially, the animal urinates through fear at leaving territory in which it is safe. Other animals then evolve a mechanism that makes use of that information. This then puts a selection pressure on the focal animal to urinate whenever it needs to make clear its territorial boundaries. As such, the evolution of a particular mechanism changed the selection pressures that act upon the species. It is likely that the emergence of culture had a similarly significant effect on human evolution, but that makes no difference to the fact that cultural transmission is a causal trigger of the expression of certain behaviors and hence should be considered a proximate mechanism.
An Example: Linguistic Structure
To make the argument clearer, we will illustrate with a specific example: the cultural evolution of linguistic structure. Many studies, mostly computational (e.g., Brighton, 2002; Hurford, 2000; Kirby, Smith, & Brighton, 2004), but more recently numerical (Smith & Kirby, 2008), mathematical (Kirby, Dowman, & Griffiths, 2007), and even experimental (Kirby, Cornish, & Smith, 2008) studies show that transmitted culture can turn unstructured and unlearnable languages into structured and learnable ones without the explicit natural selection of that structure. The typical approach within this line of research is to train an initial generation of agents4 on an unstructured language and then test how well that same generation of agents has acquired the language. Whatever is produced by that generation in the test phase is then used as the training data for the next generation, and the process is repeated. As mentioned, what often happens is that the languages come to exhibit structural properties. (This is not quite the whole story; there are important provisos and complications, but this headline result appears robust.) As such, there is the appearance of design in the final languages, despite no explicit selection for that structure. Instead, the observed structure is a consequence of the process of cultural transmission. This result has led some to claim that cultural transmission offers an alternative to natural selection as a source of design in nature; see, for example, the quotes in the previous section.
This is the wrong interpretation. Rather, this account of linguistic structure establishes the presence of cultural transmission as a possible causal trigger of the emergence of linguistic structure. As such, cultural transmission is a proximate mechanism. It is certainly not an ultimate explanation of why structured utterances are produced in the first place, as such an explanation would have to explain why the production of such utterances produces (or does not produce) inclusive fitness benefits.
Some of the misunderstanding here may have arisen because linguistic structure is a major explanandum for linguistics. Transmitted culture offers an alternative to evolutionary accounts that are based on a highly structured Universal Grammar, whereby the structure of language is an external manifestation of some innate template (e.g., Jackendoff, 1999; Pinker & Bloom, 1990). As such, it may be able to explain why language is structured in the way that it is, rather than in some other way, or indeed in no way at all (Christiansen & Chater, 2008; Kirby et al., 2007). From a biological point of view, however, this difference between a Universal Grammar and cultural transmission is a difference in the form of the proximate mechanism involved. To oversimplify horribly, is the mechanism a highly structured Universal Grammar, or is it the capacity to acquire transmitted culture? This is an interesting question that is of obvious relevance to language evolution and, indeed, linguistics and cognitive science more generally. It is not, however, a question about the ultimate evolutionary function of language. Contrary to the claims made, accounts of language structure based on transmitted culture do not challenge natural selection as the ultimate explanation of why linguistic structure exists in the first place (which is, presumably, because it makes communication easier; Pinker & Bloom, 1990). Rather, they challenge accounts based on a highly structured Universal Grammar as the proximate explanation of how that functionality is achieved.
Epigentic Inheritance as a Proximate Mechanism
The final instance of ultimate–proximate confusion we wish to highlight concerns epigenetic inheritance. The term epigenetics refers to processes that are “above” or otherwise separate to genetic change (see Holliday, 2006, for an overview). Epigenetic processes control gene expression, and sometimes this can amount to switching a gene on or off. These effects can be passed from one generation to another. Such transmission does not affect DNA code, but simply the on or off state of the gene. In recent years, epigenetic inheritance has been presented as an alternative to ultimate explanations based on natural selection5 (Jablonka & Lamb, 2005; Mameli, 2004; seeBonduriansky & Day, 2009, for a review). This view has gained some traction in the evolutionary literature on humans (e.g., Bjorkland, 2006; Fuentes, 2009; Jablonka & Lamb, 2007). In this article, we wish to highlight the fact that epigenetic inheritance is instead best understood as a proximate mechanism (see also Haig, 2007).
As an analogy, consider the debate that took part in the early part of the 20th century, when Mendelian genetics was considered a competing alternative hypothesis to natural selection (Provine, 2001). It has long been understood that this debate was mistaken, because it confused ultimate (natural selection) and mechanistic (Mendelian genetic inheritance) explanations. The same error seems to have reappeared in discussions of epigenetic inheritance. To see why, consider the following example (taken fromChampagne, 2008). When mice pups are exposed to fewer than normal interactions with their mothers, they go on to nurse and groom their own pups less. Similarly, the daughters of low-frequency grooming Long–Evans rats will in turn groom their own young less. Cross-fostering studies have demonstrated that these differences are the consequence of behavioral (rather than genetic) inheritance. In other words, the parental behavior is observed to modify the behavior of subsequent generations. The details of how this occurs do not matter to the present discussion, except to say that because this inheritance is epigenetic, it must be modulated by some proximate mechanism. (The most likely hypothesis involves dopamine release, mediated by oxytocin metabolism, which is in turn affected by prolonged exposure to stressors that may well methylate key genes; Champagne, 2008.) The consequence of this mechanism is that low grooming increases in frequency under stressful conditions, and high grooming increases in benign conditions.
Evolution and Genetics for Psychology / Daniel Nettle
This is environmentally induced plasticity, in which a proximate mechanism tracks the state of the environment and calibrates the phenotype accordingly. Consequently, the “conventional neo-Darwinist interpretation would [be to] view these epigenetic mutations as ‘switches’ evolved by natural selection to enable genes to exhibit conditional behavior” (Haig, 2007, p. 420). Such switches are “wonderful tools that increase the options available to DNA sequences but, in themselves, [they] should not challenge the beliefs of a neo-Darwinist” (Haig, 2007, p. 421). Indeed, plasticity is common in the natural work. One consequence of this flexibility in the face of ecological variation is that two genetically identical organisms can be phenotypically different by virtue of the fact that they exist in different ecologies, and hence, have differently calibrated phenotypes. Indeed, a large proportion of human diversity can be explained by just this sort of adaptation to local ecological conditions (Nettle, 2009b). For example (Nettle, 2009a), human body shape preferences correlate with the availability of food: individuals for whom the food supply is continuous and reliable tend to prefer leaner figures than those who experience food shortages. This is true between cultures (Ember, Ember, Korotyaev, & de Munck, 2005; Tovée, Swami, Furnham, & Mangalparsad, 2006;Wetsman & Marlowe, 1999), within cultures (Tovée, Furnham, & Swami, 2007), and even within individuals, who will express different preferences depending on how hungry they are (Nelson & Morrison, 2005). The difference between this case and the mice and rat examples above is the proximate mechanisms involved: epigenetic inheritance in one, and various psychological and physiological mechanisms associated with the availability of food in the other.
In short, epigenetic inheritance explains how organisms work but not why they work in that particular way. We do not deny that epigenetic inheritance is important. Instead, our point is that it is neither a challenge nor an alternative to genetic inheritance as an explanatory construct. Instead it is, like all proximate mechanisms, a different level of explanation to evolution by natural selection, which is an ultimate level process. Consequently, if epigenetics is treated as an ultimate source of explanation, conceptual confusion is likely to follow.
An additional problem is that recognition of the mechanistic character of epigenetics is also key to clarifying what needs to be done in future research. For example, if we do not recognize the different between genetic and epigenetic inheritance, we cannot address the hypothesis that epigenetic mechanisms might be adaptations. Not all organisms have epigenetic mechanisms, and the function and origin of epigenetic inheritance in humans is of interest and worthy of further research. One plausible hypothesis is that epigenetic systems are best understood as functionally (i.e., ultimately) equivalent to learning mechanisms—adaptations that enable a greater capture of ecological information than that permitted by direct coding into the genome (Dickins, in press). This hypothesis can only be explored if epigenetic inheritance is understood as a proximate phenomenon.
Concluding Remarks
The proximate–ultimate distinction follows directly from the theory of natural selection, which describes both a process (by which those biological traits associated with greater reproductive success are more likely to be inherited) and the consequences of that process (traits that appear to be designed to maximize the fitness of the organism). Thus, if we wish to explain some particular trait, we must explain both why it is associated with greater reproductive success (i.e., why it has been favored by the process of natural selection), and how it actually does that (i.e., how it works). The first of these are ultimate explanations, the latter are proximate explanations, and both are required for full biological understanding.
We have documented a number of instances in the literature in which these two types of explanations have been confused. In particular, proximate phenomena have been used to explain questions of ultimate causation. This has occurred in the literature on the evolution of cooperation, in the cultural evolutionary literature, and in discussions of epigenetics. Throughout the article, we have discussed specific reasons for these confusions and suggested ways in which confusion may be avoided in the future. Indeed, our goal here is a positive one. We do not wish to discourage evolutionary approaches to the human behavioral sciences. However, progress will be quicker and more efficiently achieved if the underlying theory, including the proximate–ultimate distinction, is properly applied.
Acknowledgments
The authors thank Max Burton-Chellow, Emma Cohen, Claire El Mouden, Andy Gardner, Daniel Nettle, and members of the Work in Progress Group at the Centre for Philosophy of Natural and Social Sciences at the London School of Economics for comments on previous drafts of this article.
Notes
- In sexually reproducing, diploid species (which includes all mammals), the coefficient of relatedness is intuitively easy to calculate: between identical twins, r = 1; between brothers and between parent and offspring, r = .5; between half-siblings, r = .25; and so on. To reflect this, Haldane once quipped that he would not give up his life to save one brother, but he would for both or for eight cousins.
- This is also why we can talk about organisms being designed for some purpose: They have been selected to behave in a way that maximizes inclusive fitness. Of course, this maximization occurs within certain constraints, such as those of history, costs, and the need for trade-offs (see Dawkins, 1982, especially Chapter 3, and Parker & Maynard Smith, 1990, for discussion).
- To avoid misunderstanding, it is worthwhile to state that we are not concerned with the dynamics of cultural evolution itself. Transmitted culture gives rise to a dynamical system that can be described as “evolutionary” in the general sense of change over time, and there is much published discussion of the dynamics of that system and how it interacts with genetic evolution. In this article, we are not concerned with either of these issues—only with how the existence of cultural transmission should be understood with respect to the ultimate–proximate distinction.
- This work has typically been studied using computational models, hence the term agents, although, as mentioned, human participants have also been used.
- Many of the issues with this claim have been discussed elsewhere (Dickins & Dickins, 2008; Haig, 2007; see also the responses to Jablonka & Lamb, 2007). For the sake of brevity, we will not enter into a general discussion of epigenetics in this article; we focus instead only on the question of whether epigenetic inheritance should be understood as an ultimate or a proximate source of explanation.
References
- Alcock J. (2009). Animal behavior. Sunderland, MA: Sinauer Associates.
- Bjorkland D.F. (2006). Mother knows best: Epigenetic inheritance, maternal effects, and the evolution of human intelligence. Developmental Review, 26, 213–242.
- Bonduriansky R., Day T. (2009). Nongenetic inheritance and its evolutionary implications. Annual Review of Ecology, Evolution, and Systematics, 40, 103–125.
- Boyd R., Richerson P.J. (1985). Culture and the evolutionary process. Chicago:University of Chicago Press.
- Brighton H. (2002). Compositional syntax from cultural transmission. Artificial Life,8, 25–54.
- Brunnander B. (2008). Is the language of intentional psychology an efficient tool for evolutionists? Studies in History and Philosophy of Biological and Biomedical Sciences, 39, 147–152.
- Champagne F.A. (2008). Epigenetic mechanisms and the transgenerational effects of maternal care. Frontiers in Neuroendocrinology, 29, 386–397.
- Christiansen M.H., Chater N. (2008). Language as shaped by the brain. Behavioral and Brain Sciences, 31, 489–558.
- Cosmides L. (1989). The logic of social exchange: Has natural selection shaped how humans reason? Studies with the Wason selection task. Cognition, 31, 187–276.
- Darwin C. (1859). The origin of species. London: John Murray.
- Dawkins R. (1979). Twelve misunderstandings of kin selection. Zeitschrift für Tierpsychologie, 51, 184–200.
- Dawkins R. (1982). The extended phenotype. Oxford, England: Oxford University Press.
- de Quervain J.-F., Fischbacher U., Treyer V., Schellhammer M., Schnyder U.,Buck A., Fehr E. (2004). The neural basis of altruistic punishment. Science, 305,1254–1258.
- de Waal F.B.M. (2006). Primates and philosophers: How morality evolved.Princeton, NJ: Princeton University Press.
- Dickins T.E. (in press). Evolutionary approaches to behaviour. In Swami V. (Ed.),Evolutionary psychology: A critical introduction. Oxford, England: Wiley-Blackwell.
- Dickins T.E., Dickins B.J.A. (2008). Mother Nature’s tolerant ways: Why non-genetic inheritance has nothing to do with evolution. New Ideas in Psychology, 26, 41–54.
- Ellingsen T., Johannesson M. (2007). Anticipated verbal feedback induces altruistic behaviour. Evolution and Human Behavior, 29, 100–105.
- Ember C.R., Ember M., Korotyaev A., de Munck V. (2005). Valuing thinness or fatness in women: Re-evaluating the effects of resource scarcity. Evolution and Human Behavior, 26, 257–270.
- Fehr E., Fischbacher U. (2003). The nature of human altruism. Nature, 425,785–791.
- Fehr E., Rockenbach B. (2004). Human altruism: Economic, neural and evolutionary perspectives. Current Opinion in Neurobiology, 14, 784–790.
- Fisher R.A. (1930). The genetical theory of natural selection. Oxford, England:Clarendon.
- Fuentes A. (2009). A new synthesis: Resituating approaches to the evolution of human behaviour. Anthropology Today, 25, 12–17.
- Gardner A. (2009). Adaptation as organism design. Biology Letters, 5, 861–864.
- Grafen A. (1985). A geometric view of relatedness. Oxford Surveys in Evolutionary Biology, 2, 28–90.
- Grafen A. (1999). Formal Darwinism: The individual-as-maximizing-agent analogy and bet-hedging. Proceedings of the Royal Society of London: Series B. Biological Sciences, 266, 799–803.
- Grafen A. (2006). Optimization of inclusive fitness. Journal of Theoretical Biology,238, 541–563.
- Grafen A. (2007). The formal Darwinism project: A mid-term report. Journal of Evolutionary Biology, 20, 1243–1254.
- Grafen A. (2009). Formalizing Darwinism and inclusive fitness theory. Philosophical Transactions of the Royal Society of London: Series B. Biological Sciences, 364,3135–3141.
- Haig D. (2007). Weismann rules! OK? Epigenetics and the Lamarckian temptation.Biology and Philosophy, 22, 415–428.
- Haldane J.B.S. (1932). The causes of evolution. New York: Harper’s.
- Hamilton W.D. (1964). The genetical evolution of social behaviour: I. Journal of Theoretical Biology, 7, 1–16.
- Hamilton W.D. (1970). Selfish and spiteful behaviour in an evolutionary model.Nature, 228, 1218–1220.
- Hamilton W.D. (1975). Innate social aptitudes of man: An approach from evolutionary genetics. In Fox R. (Ed.), Biosocial anthropology(pp. 133–153). London:Malaby Press.
- Harvey P.H., Pagel M.D. (1991). The comparative method in evolutionary biology.Oxford, England: Oxford University Press.
- Henrich J., McElreath R. (2003). The evolution of cultural evolution. Evolutionary Anthropology, 12, 123–135.
- Holden C., Mace R. (1997). Phylogenetic analysis of the evolution of lactose digestion in adults. Human Biology, 69, 605–628.
- Holliday R. (2006). Epigenetics: A historical overview. Epigenetics, 1, 76–80.
- Hurford J.R. (2000). Social transmission favours linguistic generalisation. In KnightC., Studdert-Kennedy M., Hurford J. R. (Eds.), The evolutionary emergence of language(pp. 324–352). Cambridge, MA: Cambridge University Press.
- Jablonka E., Lamb M.J. (2005). Evolution in four dimensions. Cambridge, MA: MIT Press.
- Jablonka E., Lamb M.J. (2007). Précis of evolution in four dimensions. Behavioral and Brain Sciences, 30, 353–392.
- Jackendoff R. (1999). Possible stages in the evolution of the language capacity.Trends in Cognitive Science, 3, 272–279.
- Johnson D.D.P. (2005). God’s punishment and public goods: A test of the supernatural punishment hypothesis in 186 world cultures. Human Nature, 16,410–446.
- Johnson D.D.P., Krueger O. (2004). Supernatural punishment and the evolution of cooperation. Political Theology, 5, 159–176.
- Kirby S. (2007). The evolution of language. In Dunbar R.I.M., Barrett L. (Eds.),Oxford handbook of evolutionary psychology(pp. 669–681). Oxford, England: Oxford University Press.
- Kirby S., Cornish H., Smith K. (2008). Cumulative cultural evolution in the laboratory: An experimental approach to the origins of structure in human language.Proceedings of the National Academy of Sciences, USA, 105, 10681–10686.
- Kirby S., Dowman M., Griffiths T. (2007). Innateness and culture in the evolution of language. Proceedings of the National Academy of Sciences, USA, 104, 5241–5245.
- Kirby S., Smith K., Brighton H. (2004). From UG to universals: Linguistic adaptation through iterated learning. Studies in Language, 28, 587–607.
- Krebs J.R., Davies N.B. (1993). An introduction to behavioural ecology. Oxford, England: Blackwell.
- Kuhn T.S. (1962). The structure of scientific revolutions. Chicago: University of Chicago Press.
- Mameli M. (2004). Nongenetic selection and nongenetic inheritance. British Journal for the Philosophy of Science, 55, 35–71.
- Maynard Smith J. (1964). Group selection and kin selection. Nature, 201,1145–1147.
- Maynard Smith J., Harper D.G.C. (2003). Animal signals. Oxford, England: Oxford University Press.
- Mayr E. (1963). Animal species and evolution. Cambridge, MA: Harvard University Press.
- Mesoudi A. (2009). How cultural evolutionary theory can inform social psychology, and vice versa. Psychological Review, 116, 929–952.
- Nelson L.D., Morrison E.L. (2005). The symptoms of resource scarcity: Judgements of food and finances influence preferences for potential partners. Psychological Science, 16, 167–173.
- Nettle D. (2009a). Beyond nature versus culture: Cultural variation as an evolved characteristic. Journal of the Royal Anthropological Institute, 15, 223–240.
- Nettle D. (2009b). Ecological influences on human behavioural diversity: A review of recent findings. Trends in Ecology and Evolution, 24, 618–624.
- Nettle D. (2009c). Evolution and genetics for psychology. Oxford, England: Oxford University Press.
- Parker G.A., Maynard Smith J. (1990). Optimality theory in evolutionary biology.Nature, 348, 27–33.
- Pinker S., Bloom P. (1990). Natural language and natural selection. Behavioral and Brain Sciences, 13, 707–727.
- Power C. (2009). Sexual selection models for the emergence of symbolic communication: Why they should be reversed. In Botha R.P., Knight C. (Eds.), The cradle of language(pp. 257–280). Oxford, England: Oxford University Press.
- Provine W.B. (2001). The origins of theoretical population genetics. Chicago:University of Chicago Press.
- Rendall D., Owren M.J., Ryan M.J. (2009). What do animal signals mean? Animal Behaviour, 78, 233–240.
- evolution. Chicago: University of Chicago Press.
- Scott-Phillips T.C. (2007). The social evolution of language, and the language of social evolution. Evolutionary Psychology, 5, 740–753.
- Smith K., Kirby S. (2008). Cultural evolution: Implications for understanding the human language faculty and its evolution. Philosophical Transactions of the Royal Society of London: Series B. Biological Sciences, 363, 3597–3603.
- Sober E. (1988). What is evolutionary altruism? Canadian Journal of Philosophy,14, 75–99.
- Soltis J. (2009). What do animal signals do? Animal Behaviour, 78, 1485–1486.
- Tinbergen N. (1963). On the aims and methods of ethology. Zeitschrift für Tierpsychologie, 20, 410–433.
- Tishkoff S.A., Reed F.A., Ranciaro A., Voight B.F., Babbitt C.C., Silverman J.S., et al. (2007). Convergent adaptation of human lactase persistence in Africa and Europe.Nature Genetics, 39, 31–40.
- Tomasello M. (2009). Why we cooperate. Cambridge, MA: MIT Press.
- Tovée M.J., Furnham A., Swami V. (2007). Healthy body equals beautiful body? Changing patterns of health and attractiveness with shifting socio-economic status. InFurnham A., Swami V. (Eds.), The body beautiful: Evolutionary and sociocultural perspectives(pp. 108–128). Basingstoke, England: Palgrave Macmillan.
- Tovée M.J., Swami V., Furnham A., Mangalparsad R. (2006). Changing patterns of attractiveness as observers are exposed to a different culture. Evolution and Human Behavior, 27, 443–456.
- Warneken F., Tomasello M. (2006). Altruistic helping in human infants and young chimpanzees. Science, 311, 1301–1303.
- West S.A., El Mouden C., Gardner A. (in press). 16 common misconceptions about the evolution of cooperation in humans. Evolution and Human Behavior.
- West S.A., Griffin A.S., Gardner A. (2007). Social semantics: Altruism, cooperation, mutualism and strong reciprocity. Journal of Evolutionary Biology, 20, 415–432.
- Wetsman A., Marlowe F. (1999). How universal are preferences for female waist-to-hip ratios? Evolution and Human Behavior, 20, 219–228.
- Wright S. (1931). Evolution in mendelian populations. Genetics, 16, 97–159.
- Zeifman D.M. (2001). An ethological analysis of human infant crying: Answering Tinbergen’s four questions. Developmental Psychobiology, 39, 265–285.