Medical transparency emerged from crisis and reform, transforming drug regulation from secrecy to disclosure while redefining trust, safety, and consumer responsibility in modern healthcare systems.

By Matthew A. McIntosh
Public Historian
Brewminate
Introduction: Transparency as a Modern Medical Imperative
Medical transparency, now regarded as a foundational principle of modern healthcare, is not an inherent feature of medical practice but the product of a long and contested historical process. For much of the nineteenth and early twentieth centuries, the contents, risks, and effects of medicines were often concealed from the public, leaving consumers to rely on advertising claims and trust in manufacturers. This absence of disclosure was not merely an oversight but a defining characteristic of a marketplace in which regulation was minimal and accountability limited. Consumers were expected to navigate a growing array of medical products with little reliable information, while producers operated within a system that rewarded secrecy and persuasive marketing. The eventual emergence of transparency represents a significant transformation in how medicine was understood, regulated, and consumed.
The absence of transparency in earlier periods reflected a broader commercial environment in which medicines were treated as proprietary commodities rather than regulated public goods. Patent medicine manufacturers operated with considerable freedom, marketing products that frequently contained addictive or harmful substances without disclosure. Consumers lacked both the information necessary to make informed decisions and the legal protections to challenge misleading claims. Transparency was not simply absent but structurally excluded, as secrecy functioned as a competitive advantage within an expanding consumer marketplace.
The emergence of regulatory frameworks in the twentieth century marked a decisive turning point, redefining the relationship between manufacturers, the state, and the public. Laws such as the Pure Food and Drug Act of 1906 and the Federal Food, Drug, and Cosmetic Act of 1938 introduced requirements for labeling, safety testing, and truthfulness in marketing, gradually shifting the burden of proof onto producers. These reforms were not abstract policy developments but responses to specific crises and sustained advocacy efforts, reflecting the growing expectation that medical products should be subject to oversight and accountability. Transparency was constructed through conflict, negotiation, and the accumulation of regulatory authority.
Yet the establishment of transparency did not eliminate the tensions inherent in medical regulation. Even in the modern era, the balance between disclosure, comprehension, and commercial interests remains complex and contested. Detailed labeling requirements and standardized information systems have expanded access to knowledge, but they have also introduced new challenges related to interpretation and information overload. The history of medical transparency reveals not a completed transformation but an ongoing process, in which the demand for openness continues to evolve alongside the scientific, economic, and political structures that shape healthcare.
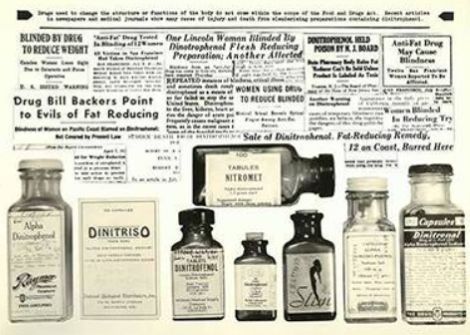
The “Buyer Beware” Era: Patent Medicines and Hidden Ingredients (Pre-1906)

Before the advent of federal regulation, the American medical marketplace operated under a principle that placed responsibility squarely on the consumer. The late nineteenth and early twentieth centuries were dominated by the widespread sale of patent medicines, products that were often marketed as cure-alls for a wide range of ailments. These remedies were sold openly in newspapers, catalogs, and traveling shows, promoted through persuasive advertising that emphasized effectiveness while omitting critical information about their contents. The absence of disclosure was not an anomaly but a defining feature of the system, leaving consumers to navigate medical choices with little reliable guidance.
The composition of these medicines frequently included substances that were not only undisclosed but potentially dangerous or addictive. Ingredients such as opium, morphine, cocaine, and high concentrations of alcohol were commonly used to produce noticeable effects, which could then be interpreted by consumers as evidence of efficacy. Because these substances could create temporary relief or stimulation, they reinforced the credibility of the product while masking underlying risks. Manufacturers deliberately exploited this dynamic, understanding that immediate, perceptible effects could generate repeat customers even if the underlying condition remained untreated. Without labeling requirements, consumers had no way of knowing what they were ingesting, and repeated use could lead not only to dependency but also to long-term physical harm. In some cases, these products were marketed to vulnerable populations, including children and the chronically ill, further amplifying the dangers associated with their widespread use.
The marketing strategies employed by patent medicine manufacturers played a central role in sustaining this system. Advertisements often featured testimonials, exaggerated claims, and appeals to authority, presenting products as scientifically validated or endorsed by medical professionals. These claims were rarely subject to verification, and the lack of regulatory oversight allowed manufacturers to craft narratives that blurred the line between medicine and entertainment. The success of these strategies depended on public trust, which was cultivated through repetition, branding, and the strategic use of emerging mass media.
The legal framework governing these products offered little protection to consumers. There were few mechanisms in place to challenge misleading claims or to hold manufacturers accountable for harm caused by their products. Courts generally treated these medicines as commercial goods rather than as substances requiring oversight, and the burden of proof fell on individuals who often lacked the resources to pursue legal action. This regulatory vacuum enabled the continued proliferation of unsafe and misrepresented remedies, reinforcing a system in which profit frequently outweighed public safety. Even when harmful effects became apparent, the lack of coordinated oversight meant that responses were slow, inconsistent, or entirely absent, allowing dangerous products to remain on the market with minimal interference.
Despite these conditions, criticism of the patent medicine industry began to grow in the late nineteenth century, driven by reformers, journalists, and medical professionals who exposed the dangers of unregulated products. Investigative reporting highlighted the hidden ingredients and deceptive practices that characterized the industry, bringing public attention to the risks that had long been obscured. These efforts played a crucial role in shaping public opinion, transforming isolated concerns into a broader movement for reform and laying the groundwork for legislative action.
The “buyer beware” era represents a critical stage in the development of modern medical regulation, illustrating the consequences of a system built on secrecy and minimal oversight. The widespread use of undisclosed and potentially harmful ingredients revealed the limits of consumer responsibility in the absence of transparency. As public awareness increased, so too did the demand for accountability, setting the stage for the regulatory transformations that would follow. The eventual shift toward federal regulation did not emerge spontaneously but was the direct result of sustained pressure from reformers and a growing recognition that unregulated markets could not adequately protect public health. The patent medicine era serves not only as a cautionary example but as the foundation upon which modern standards of medical disclosure were constructed.
The Pure Food and Drug Act of 1906: The First Step Toward Disclosure

The passage of the Pure Food and Drug Act of 1906 marked a turning point in the regulation of medical products in the United States, introducing the first meaningful federal effort to address the dangers posed by unregulated medicines. This legislation did not emerge in isolation but was the culmination of years of advocacy by reformers, scientists, and journalists who sought to expose the risks embedded within the existing marketplace. Public awareness had been shaped by investigative reporting and mounting evidence of harm, creating a political environment in which federal intervention became both possible and necessary. The Act represented an acknowledgment that consumer protection could no longer be left to individual judgment in a system structured by secrecy and misinformation.
At the center of this reform movement was Harvey Washington Wiley, chief chemist of the U.S. Department of Agriculture, whose efforts to promote food and drug safety played a critical role in shaping the legislation. Wiley and his allies argued that transparency was essential to protecting public health, emphasizing the need for accurate labeling and the disclosure of harmful ingredients. Through initiatives such as the “Poison Squad” experiments, Wiley demonstrated the dangers of unregulated additives, bringing scientific evidence into public discourse and strengthening the case for reform. His work was supported by a broader coalition that included journalists, consumer advocates, and medical professionals, all of whom contributed to increasing public pressure on lawmakers. The Act responded to these concerns by prohibiting the interstate sale of adulterated or misbranded drugs and by requiring manufacturers to disclose the presence of certain dangerous substances, including alcohol, morphine, and cocaine. While limited in scope, these provisions introduced the principle that consumers had a right to know what they were consuming, marking a fundamental shift in the relationship between producers and the public.
Despite its significance, the 1906 Act left important gaps in regulation. It did not require manufacturers to prove that their products were safe before entering the market, nor did it establish comprehensive standards for evaluating effectiveness. Enforcement mechanisms were also limited, and regulatory authority remained constrained by the legal and political realities of the time. While the Act curtailed some of the most egregious practices of the patent medicine industry, it did not eliminate the broader problem of misleading or ineffective treatments. Transparency had begun, but it remained partial and uneven.
Even with these limitations, the Act established a foundation upon which future regulatory developments would build. By introducing labeling requirements and defining misbranding as a legal violation, it created a framework for ongoing federal involvement in the oversight of medical products. The law signaled a shift away from a purely market-driven system toward one in which public health considerations played a central role. It set the stage for subsequent reforms that would expand the scope of regulation, transforming transparency from a limited requirement into a central principle of modern medical practice.
Crisis and Reform: The Elixir Sulfanilamide Disaster (1937)

The Elixir Sulfanilamide disaster of 1937 stands as one of the most consequential public health crises in the history of American drug regulation, exposing critical weaknesses in the existing legal framework. At the time, sulfanilamide was a widely used and effective antimicrobial drug, but its formulation into a liquid “elixir” introduced a fatal flaw. To dissolve the drug, the manufacturer used diethylene glycol, a toxic chemical commonly found in antifreeze, without conducting adequate safety testing. The resulting product was distributed across multiple states, leading to a wave of poisonings that revealed the absence of meaningful safeguards in the pharmaceutical market.
The human toll of the disaster was immediate and devastating. More than one hundred people, many of them children, died after ingesting the elixir, suffering severe organ failure as the toxic solvent took effect. Families and physicians initially struggled to understand the cause of the sudden and severe symptoms, as the medication had been marketed as safe and beneficial. Reports of illness emerged from multiple regions, creating confusion and delay in identifying a common source. As cases mounted, public health authorities began to connect the incidents, but by that time, the product had already reached a wide consumer base. The scale of the tragedy exposed the speed at which harmful products could circulate in an unregulated market, as well as the difficulty of responding effectively once distribution had already occurred.
At the center of the crisis was a regulatory gap embedded within the aforementioned Pure Food and Drug Act of 1906. While the law addressed misbranding and required certain disclosures, it did not mandate pre-market safety testing for drugs. The manufacturer of the elixir had technically complied with existing regulations, as the product was accurately labeled in terms of its contents and did not violate the narrow definitions of adulteration or misbranding. This legal reality shocked both the public and lawmakers, revealing that compliance with the law did not necessarily equate to safety.
The federal response to the disaster was swift and unprecedented. The U.S. Food and Drug Administration mobilized to locate and remove the product from circulation, initiating one of the first large-scale drug recalls in American history. Inspectors were dispatched across the country to trace shipments, working under significant time pressure to recover as many bottles as possible before additional harm could occur. This effort required coordination with local authorities, healthcare providers, and distributors, illustrating both the urgency of the situation and the logistical challenges involved in controlling a dispersed product. The recall process itself revealed the limitations of existing infrastructure, as there was no established system for tracking or rapidly withdrawing dangerous medications from the market. The crisis generated intense public and political pressure for reform, as the limitations of existing regulation became impossible to ignore. The tragedy transformed abstract concerns about drug safety into an urgent national issue.
In the aftermath of the disaster, the need for comprehensive regulatory change became undeniable. The Elixir Sulfanilamide case demonstrated that transparency alone was insufficient without mechanisms to ensure safety before products reached consumers. It highlighted the dangers of a system that relied on post hoc enforcement rather than preventive oversight. The crisis served as a direct catalyst for the passage of later legislation, fundamentally reshaping the regulatory landscape. The disaster marked a turning point, illustrating how public tragedy could drive structural reform and redefine the responsibilities of both manufacturers and the state.
The 1938 Federal Food, Drug, and Cosmetic Act: Safety and Labeling Modernized
The Federal Food, Drug, and Cosmetic Act of 1938 represented a fundamental transformation in the regulation of pharmaceuticals in the United States, shifting the emphasis from reactive enforcement to proactive oversight. Enacted in direct response to the Elixir Sulfanilamide disaster, the law acknowledged that existing regulatory mechanisms were insufficient to protect public health. For the first time, manufacturers were required to demonstrate that their products were safe before they could be marketed, marking a decisive break from the earlier system in which drugs could be sold without prior evaluation. This shift redefined the responsibilities of both industry and government, establishing safety as a prerequisite rather than a consequence of regulation. It also reflected a broader change in public expectations, as citizens increasingly demanded that the state play an active role in safeguarding health rather than merely responding to crises after harm had occurred. The Act signaled not only a legal adjustment but a cultural shift in how medicine, risk, and responsibility were understood in a modern industrial society.
A central feature of the 1938 Act was the expansion and clarification of labeling requirements, which aimed to ensure that consumers and medical professionals had access to accurate and meaningful information. The law strengthened provisions against misbranding, requiring that labels include adequate directions for use and truthful representations of a product’s contents and effects. While earlier legislation had introduced limited disclosure, the new framework emphasized clarity, consistency, and enforceability, recognizing that transparency was essential for informed decision-making. This development marked an important step toward standardizing how medical information was communicated to the public, reducing the ambiguity that had characterized earlier practices.
The Act also significantly broadened the authority of the FDA, enabling more effective enforcement of regulatory standards. Inspectors were granted the power to oversee manufacturing practices, monitor distribution, and take action against products deemed unsafe or misleading. This expansion of federal authority reflected a growing consensus that public health required centralized oversight capable of responding to an increasingly complex pharmaceutical landscape. By strengthening institutional capacity, the law laid the groundwork for a more systematic approach to regulation, one that could adapt to ongoing scientific and industrial developments.
Despite these advances, the 1938 Act did not resolve all challenges associated with medical transparency and safety. While it established the requirement for pre-market safety testing, it did not yet mandate proof of efficacy, leaving open the possibility that ineffective drugs could still enter the market. This limitation would later become a focal point for further reform, particularly in the wake of mid-twentieth-century pharmaceutical controversies that exposed the risks of inadequately tested treatments. Nevertheless, the legislation marked a decisive step in the evolution of modern medical regulation, embedding the principles of safety and transparency within the legal structure of the pharmaceutical industry. It created a durable institutional framework that would support subsequent expansions of regulatory authority, ensuring that the lessons of earlier crises would continue to shape policy in the decades that followed.
The Kefauver-Harris Amendments of 1962: From Safety to Efficacy

The Kefauver-Harris Amendments of 1962 marked a decisive expansion in the scope of pharmaceutical regulation, building upon the foundation established by the Act of 1938. While earlier reforms had prioritized safety and accurate labeling, the new amendments introduced a critical additional requirement: drugs must be proven effective for their intended use before receiving approval. This shift reflected growing concern that safety alone was insufficient in a rapidly evolving pharmaceutical landscape, where ineffective treatments could still be marketed and widely consumed. By mandating evidence of efficacy, the amendments fundamentally redefined the standards by which drugs were evaluated, aligning regulatory practice more closely with emerging scientific methodologies and clinical research norms. It also represented a broader intellectual shift, as policymakers and medical professionals increasingly emphasized empirical validation over tradition, anecdote, or commercial assertion. The amendments did not simply refine existing law but reoriented the entire regulatory framework toward a more evidence-driven model of medical authority.
The immediate catalyst for these reforms was the thalidomide tragedy, a global crisis in which a widely prescribed drug caused severe birth defects in thousands of children. Although the drug was never fully approved in the United States, the near-miss exposed vulnerabilities in the existing regulatory system and underscored the need for more rigorous oversight. The actions of FDA reviewer Frances Oldham Kelsey, who resisted pressure to approve the drug without sufficient data, became emblematic of the importance of strong regulatory safeguards. Public awareness of the tragedy intensified demands for reform, creating the political momentum necessary to pass comprehensive amendments that would strengthen both pre-market evaluation and ongoing monitoring.
The amendments introduced a range of new requirements designed to enhance both transparency and accountability within the pharmaceutical industry. Manufacturers were now obligated to conduct well-controlled clinical trials and to submit substantial evidence demonstrating a drug’s effectiveness. This requirement not only increased the rigor of the approval process but also standardized the methods by which medical knowledge was produced and validated. In addition, the law strengthened oversight of drug advertising, requiring that promotional materials present a balanced view of benefits and risks rather than exaggerated claims. These provisions expanded the role of scientific evidence in regulatory decision-making, ensuring that claims about medical products would be subject to verification rather than accepted at face value. Transparency was deepened, moving beyond the disclosure of ingredients to encompass the disclosure of evidence itself, thereby reshaping how both professionals and consumers understood the credibility of medical claims.
In the long term, the Kefauver-Harris Amendments reshaped the relationship between science, industry, and the state, embedding rigorous evaluation within the drug approval process. They contributed to the development of modern clinical trial standards and reinforced the authority of the FDA as a central institution in public health protection. While the increased regulatory burden also raised concerns about cost and innovation, the amendments established a new baseline for trust in medical products, ensuring that both safety and effectiveness would be subject to scrutiny. The reforms of 1962 represent a critical moment in the evolution of medical transparency, extending its reach from labeling and safety to the very evidence upon which medical claims are based.
Late Twentieth-Century Developments: Standardization and Surveillance

By the late twentieth century, the regulatory framework governing pharmaceuticals had evolved beyond the foundational concerns of safety and efficacy to encompass a more systematic approach to standardization and post-market surveillance. As the pharmaceutical industry expanded and globalized, regulators faced the challenge of ensuring not only that drugs were approved under rigorous conditions but also that their ongoing use could be monitored effectively. This period witnessed the refinement of regulatory practices, with increasing emphasis placed on consistency in labeling, clarity in communication, and the continuous collection of data on drug performance. Transparency became a dynamic process rather than a fixed requirement, extending across the entire lifecycle of a medical product.
One of the most significant developments of this era was the standardization of drug labeling, particularly for over-the-counter medications. Regulatory authorities introduced structured formats designed to present essential information in a clear and accessible manner, culminating in the familiar “Drug Facts” label. This format organized information into standardized categories, including active ingredients, uses, warnings, directions, and inactive components, thereby reducing ambiguity and improving consumer comprehension. The move toward standardized labeling reflected a recognition that transparency required not only disclosure but also intelligibility, ensuring that information could be meaningfully interpreted by a broad public. Prior to this standardization, labels often varied widely in structure and terminology, making it difficult for consumers to compare products or fully understand their risks. By imposing uniformity, regulators sought to create a shared language of medical information, one that could bridge the gap between technical knowledge and everyday decision-making. This development also aligned with broader consumer protection efforts, emphasizing that access to information must be accompanied by clarity and usability if it is to serve its intended purpose.
Advances in pharmacovigilance transformed how regulators monitored the safety of drugs after they entered the market. Systems were developed to collect and analyze reports of adverse events, allowing for the identification of risks that might not have been apparent during clinical trials. The establishment of programs such as the Food and Drug Administration’s MedWatch in 1993 created formal channels through which healthcare professionals and consumers could report side effects and complications. This shift toward surveillance acknowledged that pre-market testing, while essential, could not capture the full range of potential outcomes associated with widespread use.
These developments also reflected broader technological and institutional changes that enabled more sophisticated forms of data collection and analysis. The increasing digitization of medical records, improvements in communication networks, and the growing role of international regulatory cooperation all contributed to a more interconnected system of oversight. Regulatory agencies were no longer confined to isolated national frameworks but operated within a global environment in which information could be shared and compared across borders. This expansion of surveillance capacity enhanced the ability of regulators to respond to emerging risks, reinforcing the principle that transparency must be sustained through ongoing observation and adaptation.
Despite these advances, the late twentieth century also revealed new challenges associated with the proliferation of information. As labeling became more detailed and surveillance systems generated increasing volumes of data, questions arose regarding the accessibility and usability of this information for both professionals and the public. The tension between completeness and clarity became more pronounced, highlighting the limits of transparency when information exceeds the capacity for effective interpretation. Nevertheless, the developments of this period established a more comprehensive model of regulation, one that integrated standardization and surveillance into the broader framework of medical transparency.
The Modern Era: Transparency, Trust, and Consumer Responsibility

In the modern era, medical transparency has become both a regulatory expectation and a cultural norm, reflecting the culmination of more than a century of reform. Contemporary pharmaceutical regulation operates within a framework that assumes disclosure, standardization, and oversight as baseline requirements rather than exceptional measures. Patients are now presented with detailed information about medications, including active ingredients, potential side effects, contraindications, and proper usage instructions. This transformation represents a profound departure from earlier periods in which such information was routinely withheld, illustrating how transparency has become embedded within the structure of modern healthcare systems.
The expansion of transparency has reshaped the concept of trust within medicine, shifting it from blind reliance on authority to a more complex relationship grounded in information and verification. Patients are no longer passive recipients of treatment but active participants in healthcare decisions, expected to interpret and evaluate the information provided to them. This shift reflects broader changes in consumer culture, in which access to information is seen as a prerequisite for autonomy. It also introduces new challenges, as the responsibility for understanding complex medical data increasingly falls on individuals who may lack the necessary expertise.
Regulatory agencies have responded to these challenges by continuing to refine the presentation and accessibility of medical information. Efforts to improve labeling clarity, enhance digital access to drug data, and expand public reporting systems have sought to make transparency more functional and meaningful. Online databases, electronic health records, and mobile health applications have further expanded the availability of information, creating new avenues through which patients can engage with their own care. These digital tools have also facilitated more rapid communication of safety updates, recalls, and newly identified risks, allowing both healthcare providers and patients to respond more quickly to emerging concerns. Regulators have worked to standardize not only the content of information but also its format across digital platforms, recognizing that consistency is essential in an increasingly interconnected healthcare environment. These developments demonstrate that transparency is not a static achievement but an evolving practice shaped by technological innovation and changing expectations.
Despite these advances, tensions remain between transparency and the commercial interests of pharmaceutical companies. The disclosure of information can conflict with proprietary concerns, intellectual property protections, and marketing strategies, creating ongoing debates about the limits of openness. Regulatory frameworks must balance the need for public access to information with the incentives that drive innovation within the industry. This tension underscores the fact that transparency is not simply a technical issue but a political and economic one, embedded within broader systems of power and profit.
Moreover, the sheer volume and complexity of available information have introduced new forms of inequality in healthcare. While transparency aims to empower consumers, it can also disadvantage those who lack the resources, education, or time to fully engage with detailed medical data. The expectation that individuals will navigate complex information systems can create disparities in outcomes, as more informed or supported patients are better positioned to benefit from available knowledge. Transparency does not automatically produce equity, and its effectiveness depends on the broader social and institutional context in which it operates.
The modern era of medical transparency reflects both achievement and limitation, revealing the ongoing interplay between knowledge, authority, and responsibility in healthcare. While regulatory frameworks have succeeded in establishing disclosure as a fundamental principle, the interpretation and use of that information remain uneven and contested. Transparency has transformed the relationship between patients, providers, and manufacturers, but it has not eliminated the challenges associated with complexity, inequality, and competing interests. In many ways, the success of transparency has generated new expectations that extend beyond disclosure itself, including demands for clarity, accessibility, and meaningful engagement with information. As healthcare systems continue to evolve, these expectations will likely intensify, requiring ongoing adaptation from regulators, providers, and industry alike. The pursuit of transparency continues, shaped by evolving expectations and the persistent need to balance openness with practicality in an increasingly complex medical landscape.
Historiography: Interpreting the Rise of Medical Transparency
Historians have interpreted the rise of medical transparency through a variety of analytical frameworks, often emphasizing the interplay between state power, professional authority, and consumer protection. Early scholarship tended to frame regulatory developments as a progressive narrative, in which scientific advancement and public advocacy gradually produced a more rational and humane system of oversight. Within this interpretation, legislation such as the Pure Food and Drug Act of 1906 and the Federal Food, Drug, and Cosmetic Act of 1938 represented milestones in a linear movement toward greater accountability and safety. Transparency, in this view, emerged as a natural extension of modernization, reflecting the growing capacity of institutions to regulate complex industries.
More recent historiography has challenged this straightforward narrative by highlighting the contested and uneven nature of regulatory change. Scholars have emphasized the role of institutional reputation and political negotiation in shaping the development of agencies like the FDA. Rather than viewing transparency as an inevitable outcome of progress, this perspective situates it within a landscape of competing interests, in which regulatory authority had to be constructed, defended, and expanded over time. These scholars point to the importance of crisis moments, such as the Elixir Sulfanilamide disaster and the thalidomide scare, not as isolated turning points but as opportunities that reformers used to push for expanded authority. In this reading, transparency emerges from strategic action within bureaucratic and political systems, rather than from a simple accumulation of knowledge or goodwill. The development of regulatory credibility itself becomes central, as agencies had to build trust with both the public and policymakers to enforce increasingly complex standards.
Another strand of scholarship has focused on the cultural and economic dimensions of medical transparency, examining how consumer expectations and market dynamics influenced regulatory developments. Historians of consumer culture have argued that the demand for information about medical products was closely tied to broader transformations in advertising, mass media, and public trust. The rise of standardized labeling and disclosure requirements can be understood not only as legal innovations but also as responses to changing perceptions of risk and responsibility within a consumer society. In this interpretation, transparency reflects a shift in the relationship between producers and consumers, mediated by both cultural norms and economic pressures.
Critical perspectives have drawn attention to the limitations and ambiguities of transparency as a regulatory goal. Some scholars argue that the emphasis on disclosure can obscure deeper structural issues, such as inequality in access to healthcare and the persistent influence of corporate interests. From this viewpoint, transparency does not necessarily resolve the tensions inherent in pharmaceutical regulation but may instead reconfigure them, shifting responsibility onto consumers without fully addressing underlying disparities. Critics also note that the sheer volume of information provided through modern labeling and reporting systems can overwhelm rather than empower, particularly for individuals without specialized knowledge. Transparency may function more as a formal compliance mechanism than as a substantive guarantee of understanding, raising questions about whether disclosure alone can achieve the goals it is intended to serve. This line of analysis complicates the assumption that more information automatically leads to better outcomes, suggesting that the effectiveness of transparency depends on broader social and institutional conditions.
These historiographical approaches reveal that the rise of medical transparency is best understood as a multifaceted and contested process rather than a singular achievement. It reflects the convergence of scientific, political, economic, and cultural forces, each contributing to the evolving framework of regulation. By examining these differing interpretations, historians have demonstrated that transparency is not simply a technical solution to the problem of information asymmetry but a concept shaped by ongoing debates about authority, responsibility, and the role of the state. As such, the historiography of medical transparency continues to evolve, offering new insights into the complexities of modern healthcare systems.
Conclusion: From Secrecy to Disclosure, and Its Limits
The historical trajectory of medical transparency reveals a profound transformation in the relationship between knowledge, power, and public health. What began as a marketplace defined by secrecy, misleading claims, and minimal oversight gradually evolved into a regulatory system grounded in disclosure, verification, and institutional accountability. This shift was neither inevitable nor linear, but rather the product of repeated crises, sustained advocacy, and the growing recognition that unregulated medical markets posed significant risks to society. Transparency emerged not merely as a policy tool but as a defining principle of modern healthcare, reshaping expectations about what patients are entitled to know. It also reconfigured the authority of both medical professionals and regulatory institutions, embedding the expectation that claims must be supported by evidence and made accessible to scrutiny. The evolution of transparency reflects a broader transformation in modern governance, in which knowledge itself becomes a central instrument of regulation and public trust.
The expansion of transparency has fundamentally altered the role of the consumer within the medical system. Individuals are now expected to engage actively with detailed information about medications, weighing risks and benefits in ways that were once the exclusive domain of professionals. This transformation reflects broader cultural shifts toward autonomy and informed decision-making, but it also introduces new responsibilities that are unevenly distributed across populations. The availability of information does not guarantee comprehension, and the burden of interpretation can fall most heavily on those least equipped to manage it, revealing the limits of transparency as a universal solution.
Moreover, the history of medical transparency underscores the persistent tension between openness and competing interests within the pharmaceutical industry. While regulatory frameworks have succeeded in mandating disclosure and standardizing information, they must continually navigate the competing demands of innovation, profit, and public safety. Transparency can expose risks and reduce fraud, but it cannot fully eliminate the structural dynamics that shape how medical products are developed, marketed, and consumed. Pharmaceutical companies continue to operate within competitive markets that reward innovation but also incentivize strategic communication, selective emphasis, and the protection of proprietary information. Regulators must constantly balance the need for public access to information with the realities of industrial research and development, ensuring that disclosure requirements do not undermine the incentives that drive medical advancement. This ongoing negotiation highlights the fact that transparency is not a fixed endpoint but a continually evolving compromise shaped by economic, political, and scientific considerations.
Ultimately, the movement from secrecy to disclosure represents a significant achievement, but one that remains incomplete and contested. Transparency has transformed the foundations of medical regulation, making information more accessible and accountability more visible, yet it has not resolved all of the challenges inherent in modern healthcare. The complexity of medical knowledge, the persistence of inequality, and the influence of economic incentives continue to shape how transparency functions in practice. The history of medical transparency is not a closed narrative but an evolving on
Bibliography
- Alizadeh, Majid, Nazila Azizi, Samireh Mahdavi, and Fouad Baghlani. “Unveiling the Shadows: Obstacles, Consequences, and Challenges of Information Opacity in Healthcare Systems.” Philosophy, Ethics, and Humanities in Medicine 20:6 (2025).
- Ballentine, Carol. “Taste of Raspberries, Taste of Death: The 1937 Elixir Sulfanilamide Incident.” FDA Consumer 1981.
- Brody, Howard. “Transparency: Informed Consent in Primary Care.” The Hastings Center Report 19:5 (1989), 5-9.
- Carpenter, Daniel. Reputation and Power: Organizational Image and Pharmaceutical Regulation at the FDA. Princeton: Princeton University Press, 2010.
- Greene, Jeremy A. Generic: The Unbranding of Modern Medicine. Baltimore: Johns Hopkins University Press, 2014.
- Mehrotra, Ateev, Tyler Brannen, and Anna D. Sinaiko. “Use Patterns of a State Health Care Price Transparency Web Site: What Do Patients Shop For?” Inquiry 51:1 (2014), 1-3.
- Szumigalski, Kristoffer. “Why We Need Price Transparency in Healthcare.” Harvard Public Health Review 22 (2019), 1-4.
- Temin, Peter. Taking Your Medicine: Drug Regulation in the United States. Cambridge, MA: Harvard University Press, 1980.
- U.S. Food and Drug Administration. “The 1906 Food and Drugs Act and Its Enforcement.” Accessed 2026.
- —-. “Drug Labeling and Patient Information.” Accessed 2026.
- —-. “Elixir Sulfanilamide Disaster.” Accessed 2026.
- —-. “Federal Food, Drug, and Cosmetic Act (1938).” Accessed 2026.
- —-. “MedWatch: The FDA Safety Information and Adverse Event Reporting Program.” Accessed 2026.
- —-. “Milestones in U.S. Food and Drug Law.” Accessed 2026.
- Young, James Harvey. The Medical Messiahs: A Social History of Health Quackery in Twentieth-Century America. Princeton: Princeton University Press, 1967.
- —-. The Toadstool Millionaires: A Social History of Patent Medicines in America before Federal Regulation. Princeton: Princeton University Press, 1961.
Originally published by Brewminate, 04.01.2026, under the terms of a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International license.