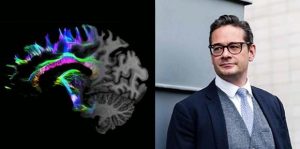
Professor Richard Gilbertson from the CRUK Cambridge Institute / Photo by Carol Sachs
Thirty years ago, Professor Richard Gilbertson pledged to implement a 15 per cent reduction in mortality from children’s brain cancer. This is the story of what happened next.
12.14.2016
On the children’s ward at Newcastle General Hospital in 1986, medical student Richard Gilbertson got his first taste of life as a paediatric oncologist. He looked around the ward and saw a child in a bed, in a dark corner. “She has a medulloblastoma that has returned,” the consultant said. “What can we do for her?” asked Gilbertson, who had been fascinated by medulloblastomas – one of the commonest malignant brain tumours in children – since his first year of medicine, when he was randomly assigned to do a project on them. “Nothing,” the consultant replied. “The only thing we can do is let her die in peace.”
“I got so angry,” remembers Gilbertson – now Professor – sitting in his airy office on the first floor of the vast glass-and-steel Cancer Research UK Cambridge Institute (CRUK CI) at the Li Ka Shing Centre. “It was the 1980s and there was nothing we could do for a child with a brain tumour. That was completely unacceptable to me. And I know it sounds contrived, but I made up my mind from that moment that I was going to do something.”
That ‘something’ was initially a number dreamed up in a Newcastle pub: relaxing with a beer after a long week, Gilbertson and his fellow medical students decided that by the time they retired, they should take personal responsibility for implementing a 15 per cent reduction in mortality, from whatever disease they chose.
Gilbertson never forgot that pledge and, 30 years later, you could argue that he’s fulfilled it. As a result of his insights into how children’s brain tumours behave, more children than ever before are surviving them. However this, he argues, is because of better patient care and understanding of the condition, not more effective treatments, earlier detection or prevention. These are the things, he says, that will fulfil his latest ambition: a world without cancer.
When Gilbertson began studying brain tumours, they were all regarded as the same disease, and needing the same treatment. He proved that the main types of tumour not only behaved differently from each other but are also totally different diseases.
“All tumours generate cells, and these cells speak different languages – express different genes – in the same way that we all speak with an accent,” he explains. “So say we had a French person, an Irish person, and a Chinese person who all spoke English, we could identify their country of origin from their accent. We thought: if we can first identify the ‘accent’ that normal cells in the brain speak with and compare them with the ‘accent’ of the tumour cells, we can trace the origin of those cells back – just like we hear a Cockney speak and we know he’s from London.”
When Gilbertson and his team studied cancer ‘accents’ in brain tumours, they fell very clearly into four different categories – four different diseases arising from four different cell types. One tumour has blood vessels like sieves, for example. It’s an achilles heel that doctors can exploit – chemotherapy drugs in the bloodstream can permeate the tumour far more efficiently.
The World Health Organisation (WHO) has now adopted Gilbertson’s classification, and children around the world now receive treatment matched to their category of tumour.
All this was achieved while Gilbertson was Cancer Centre and Scientific Director at St Jude Children’s Research Hospital, Memphis, one of the world’s leading children’s cancer hospitals, where he worked for 15 years. Yet his research seemed to be taking him more and more in the direction of not just paediatric cancer but the entirety of cancer. “Of course, they are different diseases. But I firmly believe that cancer needs to be thought of as a continuum. What you’re looking at is how development goes wrong, whether you’re seven or 70.”
In 2015, Gilbertson was appointed as Li Ka Shing Chair of Oncology in Cambridge and Director of the Cambridge Cancer Centre, with access to both child and adult cancers and, he says, “the best minds in the world”. These minds – physicists, engineers, chemists – are, right now, working out how Gilbertson’s dream of a cancer-free world can become reality.
There are big ideas to be worked on. “One of the things that’s always puzzled me as a paediatric oncologist is: why don’t children get cancer more?” Gilbertson says. “After all, as children grow, they experience massive cell proliferation. Cancer happens when a cell’s DNA goes wrong. Think of them as accelerators that make cells divide too much, with no brakes. Yet paediatric cancers are quite rare – much more so than adult cancer. People say it’s because children don’t smoke or live for 70 years or do those things which cause mistakes in a cell’s DNA. I don’t buy that. I think it’s partly true. But there must be something that protects children in the design of their cells from actually getting cancer.”
Gilbertson and his team have just completed a seven-year study identifying the cells that make cancers in children and adults. When his researchers challenged healthy cells with the mutations that drive cancer, they found that children’s stem cells appeared to be intrinsically resistant.
There was something about them that stopped them making cancer – unlike the adult cells, which weren’t resistant. “That’s terribly exciting, because if I can look into a child’s cells and work out the biology that is protecting that child’s cells from cancer, then maybe we could reproduce that in an adult cell with a drug. And if you do that, you’ve got a preventative for cancer. We are working on that right now, and it’s something I will be pursuing.”
All possibilities are explored. Gilbertson’s lab is currently screening around 1.2 million compounds, found everywhere from the depths of the Amazonian rainforest to the bottom of the ocean. Out of these, four have possibilities as potential treatments and are being developed. Then there are the drugs that already exist, that have been shown to be effective against other cancers: these are being screened as well. Taking a chemical compound from a tree, crushing it up and putting it on cancerous cells is one avenue of exploration, to be sure. But making that compound into a drug that a patient can actually take is a very long process. It’s far quicker to take a drug that already exists and give it to that child with the brain tumour. “It’s a bit like when your roof is leaking and you put a saucepan on the floor,” says Gilbertson. “The saucepan wasn’t designed to do that. It was designed to cook carrots. But it’s actually useful to catch water in.”
Innovations around early diagnosis are also being examined. Just 25 per cent of people who are diagnosed with one of the eight most common cancers in the late stages will be alive 10 years later. Diagnose the same cancers just a few months earlier, when the disease is in its early stages, and 80 per cent of those patients will be alive in 10 years. The earlier you diagnose, the better chance of a cure. And here, again, it’s all about understanding the tumour, working out its strengths and its weaknesses, finding the things it does that can be turned against it.
Gilbertson points to the work of Professor Rebecca Fitzgerald at the MRC Cancer Unit, a partner of the Cancer Centre, as a perfect example. She developed the cytosponge – the equivalent of a cervical smear for the oesophagus, a notoriously hard area in which to spot pre-cancerous changes. It’s a tiny pill containing an even tinier sponge on a ‘fishing line’. The patient swallows it, the pill hits the stomach and dissolves, leaving the sponge behind. The line is then pulled up, with the sponge scraping a cell sample from the oesophagus on the way up. “What we will see increasingly in cancer is a push towards diagnosing early, and people becoming increasingly used to going through their GPs,” he says. “If you asked my dad’s generation if they had their blood pressure checked regularly, they would say no. Those kind of early diagnostics didn’t exist then but now they do, and are common practice. I think you will see that in cancer.”
Yesterday, Gilbertson mentions, he was meeting with the inventor of a breath tester to detect lung cancer. Tumour cells have a different way of consuming food than normal cells, he explains, so they produce slightly different waste products. Some of these are volatile, and these tell-tale compounds will appear in the breath – so they can be detected. The team are also developing tests for circulating tumour DNA. It’s now known that DNA isn’t present just in cells: it floats around the bloodstream. Tumours are caused by mutations in that DNA: if you create a blood test sensitive enough to detect those mistakes, you could identify that tumour before the person even starts to show symptoms.
Imagine, he says, the conversations around cancer in the future. “If people never get cancer as we know it, they’ll be saying: ‘Oh, I’ve been diagnosed with gastric cancer but the doctor’s just fixed it.’ Imagine a world where well-person clinics test accurately for the earliest cancers every year, rather than patients walking around with tumours inside them for years on end – and only when they get ill do we do something about it. Imagine a child going into a clinic for a five-year checkup, and having a blood test which reveals she has cancer. You intervene with a relatively non-toxic treatment – even minor surgery – and that’s it.” A world without cancer, where the dark corners of the ward are banished to the history books. It’s a pledge worth pursuing.