Flickr, Creative Commons
By Dr. Sharon Nell Dewitte (left) and Dr. Maryanne Kowaleski (right)
Dewitte: Associate Professor of Anthropology, University of South Carolina
Kowaleski: Joseph Fitzpatrick SJ Distinguished Professor of History and Medieval Studies, Fordham University
Abstract
The fourteenth-century Black Death was one of the most important and devastating epidemics in human history. It caused or accelerated important demographic, economic, political, and social changes throughout the Old World and has therefore been the subject of scholarly research in a variety of fields, including history, anthropology, demography, and molecular biology. In this paper, we examine the Black Death (specifically, the first and second outbreaks of fourteenth-century plague, c. 1347–1351 and 1361–1362) from bioarchaeological and historical perspectives, focusing on attempts to reconstruct mortality patterns and addressing the questions: Who died in England during the Black Death? How did they die, where and when? We evaluate how historical and bioarchaeological sources are uniquely informative about these questions and highlight the limitations that are associated with each type of data. The combination of the two bodies of evidence, when possible, can provide insights that are not possible when each is analyzed in isolation.
Introduction
The fourteenth-century Black Death was one of the most important epidemics in human history, as it caused or accelerated important demographic, economic, political, and social changes throughout the Old World. This epidemic was devastating not only because of its extremely high mortality levels, but also because the deaths it caused were concentrated within a brief period. In the four years that the Black Death was at its height in Europe (between 1347 and 1351), the epidemic killed 30 to 60 percent of the population, amounting to tens of millions of people. This essay argues that a full understanding of the epidemiology of this devastating plague—that is, its levels of mortality; the distribution of deaths by age, sex, wealth, and location; and the susceptibility of victims to disease—can only be attained by bringing together archaeological and documentary evidence. We focus on the people who succumbed to the Black Death in medieval England and make a deliberate effort to compare and contrast not only what the different types of documentary and material evidence say, but also the analytic questions and methodological approaches that different disciplines have adopted to discuss plague during the fourteenth century, particularly the first (1348–1349) and second (1361–1362) waves of the Black Death.
The Black Death is increasingly recognized as a semi-global phenomenon, likely starting in East Asia and spreading throughout Central Asia, Europe, and North Africa.[1] It is often described as the second of three known pandemics caused by Yersinia pestis, the bacterium associated with plague. According to this view, the First Pandemic was the Plague of Justinian, which began in the sixth century C.E., with recurrent outbreaks for about two centuries thereafter.[2] The Black Death of 1347–1351 and subsequent visitations of the plague, including the Great Plague that hit London in 1665, constituted the Second Pandemic. The Third Pandemic began in China in the late eighteenth century, spread from there throughout much of the world, and ended by the mid-twentieth century.[3] Outbreaks of plague continue to this day, although modern antibiotics and public health measures have greatly reduced the mortality rate and spread of the disease. Scholarly disagreement about whether the Black Death was the same bubonic plague of the Third Pandemic or some other disease has raged for some time, but the recent discovery of Y. pestis DNA in the dental pulp of victims of the mid-fourteenth-century plague has settled this question.[4] We are still unable to explain fully, however, the different clinical manifestations and epidemiological patterns of Y. pestis, including the significantly higher mortality and faster spread of medieval plague, and the likelihood that medieval plague did not follow the same transmission path from animal host to flea to humans often identified in modern bubonic plague.
The documentary sources employed by historians to analyze the Black Death have been known for some time. They include chronicles, which offer direct statements from contemporaries about the symptoms, pace, and mortality rates of the plague, as well as appointments of parish priests, wills, and especially manorial records, which reflect the experience of the rural majority. In the last twenty-five years, a new source of material evidence has become available. Bioarchaeology investigates human skeletal remains excavated from archaeological sites to determine the ages at which people died, as well as their sex, health, diet, migration, experience of interpersonal violence, and other behaviors. The bioarchaeological analysis of skeletal remains is vital for a complete understanding of life in the past because it can yield data often missing from historical documents and can be employed to test assumptions and inferences based on documentary evidence.[5]
How the Black Death Killed: Symptoms, Spread, and Duration
Most of what we know about the symptoms of the Black Death comes from medieval chroniclers, many of them eyewitnesses, and from plague treatises written years and often decades after the first outbreak of plague. Their descriptions are broadly similar, pointing to fever, headache, lethargy, dark patches or small black pustules on the skin, and buboes or swollen lymph glands in the armpit, groin, or neck. Other symptoms included nausea, diarrhea, vomiting, and the coughing up of blood.[6] In The Decameron, Boccaccio describes the “plague-boils” of the Black Death as “certain swellings, either in the groin or under the armpits, whereof some waxed of the bigness of a common apple, others like unto an egg, some more and some less.”[7] The English chronicler Geoffrey le Baker wrote that victims “were tormented by boils which broke out suddenly in various parts of the body, and were so hard and dry that when they were lanced hardly any liquid flowed out. . . . Other victims had little black pustules scattered over the skin of the whole body.”[8] Medieval observers stressed how quickly the disease progressed, noting that death usually came within three days.[9]
Scientists have identified several clinical forms of plague in humans: bubonic, septicemic, pneumonic, gastrointestinal, and pharyngeal.[10] The bubonic form is predominant in modern outbreaks of plague. Its main symptoms are exactly those described by medieval sources: headache, chills, nausea, and fever followed by hard and increasingly painful swellings near lymph nodes in the neck behind the ears, armpits, and groin.[11] Septicemic and pneumonic plague usually develop secondarily to bubonic plague, but can rarely occur as primary diseases if the plague bacillus infects the bloodstream or lungs, respectively, without first infecting the lymphatic system.[12] All forms of plague are different clinical manifestations of infection with Y. pestis, and the proportions of infected individuals who die from different forms of untreated plague are high, ranging from 30 to 60 percent for bubonic and septicemic plague and approaching 100 percent for pneumonic plague.[13]
Several medieval accounts recognized the distinct symptoms of pneumonic plague, which included respiratory distress such as shortage of breath and coughing up of blood or sputum, as well as a quick death.[14] To explain the apparent greater virulence of historic plague compared to modern plague, some scholars have suggested that the Black Death was an outbreak of pneumonic plague, which has very high case-fatality rates and can spread from person to person.[15] However, although pneumonic plague can develop in an area after bubonic plague has taken hold,[16] evidence from the pre-antibiotic era suggests that most primary cases fail to transmit the disease to other people.[17] Fatality rates for those who contract pneumonic plague are very high, but during a modern pre-antibiotic outbreak of bubonic plague, pneumonic plague accounted for only a small fraction of total cases.[18] At present, therefore, it is not clear whether attributing the Black Death to pneumonic plague satisfactorily explains the patterns of the medieval epidemic.
Medieval documentary sources also offer information on the spread, seasonality, and duration of the disease. Chroniclers identified the first English victims during the summer of 1348 in port towns, emphasizing its arrival on ships from the Continent, though they disagree on the exact dates (sometime between late June and late September) and which ports it struck first (Bristol, Southampton, or Melcombe).[19] Some chroniclers offer more precise trajectories for individual locales, as did Robert of Avesbury, a London clerk who wrote that the plague arrived in London on All Saints Day (1 November 1348), was especially virulent from Candelmas (2 February) to Easter (12 April), and ended around Pentecost (31 May 1349).[20] A few medieval observers venture an estimate of the Black Death’s duration in England in noting that the 1348–1349 plague “lasted for a whole year” in England although at least one tracked it at two years.[21] There were no such estimates by chroniclers for subsequent visitations of the plague in fourteenth-century England.[22]
Lists kept in bishop’s registers recording the appointments of parish priests to newly vacant posts are a more reliable source for studying the chronological and geographical distribution of the Black Death in England. The dates and location of these “institutions to benefices,” offer a rough guide to the pace and spread of the plague, indicating that around 45 percent of the beneficed parish clergy died in the course of just over twelve months.[23] Various problems, particularly in accounting for the lag between the incumbent’s actual death (which is rarely known) and the appointment of a new priest, mean that these studies tend to underestimate the speed at which the Black Death spread.[24] The timing of the plague in particular villages can also be discerned in scattered references to the “pestilence” in the dated court rolls of specific manors, which sometimes contain countable references to the large numbers of heirs who came to court to claim properties from deceased tenants.[25] Historians employ wills to date when the plague raged in specific towns, such as London, though here too the lag between the actual death of the will-maker and the enrollment of the will can skew estimates of the progression of the disease, while the exclusion of all but the wealthy who left wills narrows the cohort to a very small and biased sample of the population.[26]
Figure 1: The spread of the Black Death in England, 1348-1349. The dark shading indicates when the Black Death was prevalent in a region; light shading marks areas where the plague had waned; no shading points to areas to which the plague had yet to spread. From The Scourging Angel: The Black Death in the British Isles, by Benedict Gummer (The Bodley Head and Vintage, 2009). Reprinted by permission of The Random House Group Limited.
Despite their drawbacks, these sources can provide considerable data on when and exactly where the Black Death raged, phenomena that have largely been inaccessible to bioarchaeologists and archaeologists, though recent work that counts pottery fragment as a proxy for human demographic trends represents a promising approach.[27] By combining evidence from a range of complementary records, such as has been done recently for several towns and regions in medieval England, scholars can offer a fairly comprehensive outline of the chronology of the 1348–1349 epidemic.[28] The documentary sources agree that the Black Death in fourteenth-century England was most virulent in the summer months; outbreaks of modern bubonic plague also show marked seasonality.[29] These sources also indicate the speed at which the Black Death spread in England, taking about a year and a half to cover the length of the country and averaging about 1 km per day, a pace possible because plague probably entered the British Isles via several different seaports from where it spread inland (Figure 1).[30] In contrast, modern bubonic plague moved much more slowly. It took over one hundred years for bubonic plague to spread from Yunnan Province in southwest China to Hong Kong in the late eighteenth and nineteenth centuries.[31]
This contrast raises questions about how plague was transmitted in the medieval period. Medieval chroniclers such as Baker and others articulated a fear of contagion; they believed that the disease could be spread merely by touching the clothing or bedding of the sick, going so far as to declare that anyone “who touched the dead or the sick were immediately infected themselves and died.”[32] Medieval people, however, had no understanding of how the disease was contracted or transmitted by different vectors (the organisms such as fleas or ticks through which the disease was carried). Until recently, most scientists have assumed that medieval plague was transmitted in the same fashion as modern bubonic plague, which is a zoonosis, a disease among animals spread by an insect vector.[33] Y. pestis is an obligate parasite that can reproduce only by exploiting its mammalian or insect host, so bubonic plague in humans is dependent upon the cyclic transmission between animal hosts and the ectoparasites that live and feed on them.[34] Humans often fall ill when they come into contact with infected animals and are bitten by the animal’s fleas or other ectoparasites. This dependence on animals for transmission has been used to explain why modern bubonic plague spread so slowly, but ignores the potential effect of human transportation of infected animals on how quickly and widely the disease spread. It also assumes that plague is primarily spread by rats, an assumption undermined by the biased nature of the investigations of early twentieth-century plague in India that have influenced studies since, and more recent evidence that over 200 animals can serve as hosts for plague.[35] Recent scientific research has shown that Y. pestis can be transmitted to humans via not only the rat flea, but also by other insect vectors such as the cat flea, human flea, human body louse, and human hair louse.[36] The application of mathematical models of plague transmission suggests that lice may have been involved in the spread of the disease during the Black Death.[37] In other words, much of what people read about the epidemiology of the Black Death—particularly that it was spread by rats and fleas—has not been conclusively established using evidence from the medieval period but rather reflects the imposition of a rat-centric view of plague dynamics that is problematic even for modern plague.[38] Further work needs to be done to clarify the transmission dynamics during the medieval period.
How Many the Black Death Killed
Table 1: Mortality rates of English rural tenants (mostly men), 1348–1349
| Manors | County | Mortality rate | Source type | Source |
|---|---|---|---|---|
| SOUTH-WEST | ||||
| Calstock | Corn. | 42% | S: Heriots | Hatcher, 105 |
| Climsland | Corn. | 62% | S: Heriots | Hatcher, 105 |
| Marnhull | Dorset | 36% | M: Head-tax (LL) | Ecclestone, 26 |
| Ashcott | Soms. | 44% | M: Head-tax (LL) | Thompson, 168 |
| Batcombe | Soms. | 54% | M: Head-tax (LL) | Ecclestone, 26 |
| Ditcheat | Soms. | 54% | M: Head-tax (LL) | Ecclestone, 26 |
| Greinton | Soms. | 47% | M: Head-tax (LL) | Thompson, 168 |
| Ham | Soms. | 42% | M: Head-tax (LL) | Ecclestone, 26 |
| Mells | Soms. | 58% | M: Head-tax (LL) | Ecclestone, 26 |
| Pilton | Soms. | 61% | M: Head-tax (LL) | Ecclestone, 26 |
| Street | Soms. | 55% | M: Head-tax (LL) | Thompson, 168 |
| Walton | Soms. | 52% | M: Head-tax (LL) | Thompson, 168 |
| Walton | Soms. | 61% | M: Head-tax (LL) | Ecclestone, 26 |
| Badbury | Wilts. | 76% | M: Head-tax (LL) | Ecclestone, 26 |
| Christian Malford | Wilts. | 66% | M: Head-tax (LL) | Ecclestone, 26 |
| Damerham | Wilts. | 64% | M: Head-tax (LL) | Ecclestone, 26 |
| Downton | Wilts. | 66% | E: Heriots (incl. some LL) | Ballard, 213 |
| Grittleton | Wilts. | 63% | M: Head-tax (LL) | Ecclestone, 26 |
| Idmiston | Wilts. | 50% | M: Head-tax (LL) | Ecclestone, 26 |
| Kington | Wilts. | 56% | M: Head-tax (LL) | Ecclestone, 26 |
| Nettleton | Wilts. | 48% | M: Head-tax (LL) | Ecclestone, 26 |
| SOUTH-EAST | ||||
| Alresford | Hants. | 59% | M. Heriots | Arthur, 61 |
| Alverstoke | Hants. | 95% | M. Heriots | Arthur, 61 |
| Ashmanworth | Hants. | 71% | M. Heriots | Arthur, 61 |
| Beauworth | Hants. | 80% | M. Heriots | Arthur, 61 |
| Bishop’s Waltham | Hants. | 65% | E: Heriots | Titow, 70 |
| Cadland | Hants. | 100% | M: Heriots | Watts, 27 |
| Cheriton | Hants. | 63% | M. Heriots | Arthur, 61 |
| Corhampton | Hants. | 61% | M: Heriots | Watts, 27 |
| Crawley | Hants. | 71% | M. Heriots | Arthur, 61 |
| Ecchinswell | Hants. | 68% | M. Heriots | Arthur, 61 |
| Funtley | Hants. | 100% | M: Heriots | Watts, 27 |
| Gosport | Hants. | 100% | M. Heriots | Arthur, 61 |
| Hambledon | Hants. | 94% | M. Heriots | Arthur, 61 |
| North Waltham | Hants. | 54% | M. Heriots | Arthur, 61 |
| Stubbington | Hants. | 41% | M: Heriots | Watts, 27 |
| Swanwick | Hants. | 64% | M: Heriots | Watts, 27 |
| Titchfield | Hants. | 72% | M: Heriots | Watts, 27 |
| Wallsworth | Hants. | 62% | M: Heriots | Watts, 27 |
| Woodhay | Hants. | 82% | M. Heriots | Arthur, 61 |
| EAST | ||||
| Cottenham | Cambs. | 49% | M: Heriots | Ravensdale, 198 |
| Oakington | Cambs. | 64% | M: Heriots | Page, 121 |
| Dry Drayton | Cambs. | 48% | M: Heriots | Page, 121 |
| Chatham Hall | Essex | 45% | S: Tithing penny | Poos, Rural Society, 107n20. |
| Fingrith | Essex | 63% | S: Heriots | Fisher, 13–20 |
| Great Waltham | Essex | 44% | S: Tithing penny | Poos, Rural Society, 107n20 |
| High Easter | Essex | 54% | S: Tithing penny | Poos, Rural Society, 107n20 |
| Margaret Roding | Essex | 26% | S: Tithing penny | Poos, Rural Society, 107n20 |
| Hakeford Hall | Norf. | 56% | S: Heriots | Campbell, “Population Pressure,” 96 |
| Walsham-le-Willows | Suff. | 45–55% | S: Heriots | Lock, 320–01 |
| MIDLANDS | ||||
| Ashbury | Berks. | 55% | M: Head-tax (LL) | Ecclestone, 26 |
| Buckland | Berks. | 60% | M: Head-tax (LL) | Ecclestone, 26 |
| Brightwell | Berks. | 29% | E: Heriots | Ballard, 207–08 |
| Kibworth Beauchamp | Leic. | 67% | S: Tithing penny | Postles, 47 |
| Kibworth Harcourt | Leic. | 40% | C: Tithing penny | Postles, 46 |
| Witney | Oxon. | 66 | E: Heriots | Ballard, 195–96, 208 |
| Cuxham | Oxon. | 65% | C: Heriots | Harvey, 135 |
| Higham Ferrers | Northm. | 57% | S: Court roll list | Groome, 310 |
| Halesowen | Worc. | 40% | M: Entry fines | Razi, 103 |
| NORTH | ||||
| Aycliffe | Durham | 61% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Billingham | Durham | 45% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Burdon | Durham | 64% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Dalton-le-Dale | Durham | 69% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| East Rainton | Durham | 29% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Fulwell | Durham | 56% | M: Heriots | R. Lomas, 129 |
| Harton | Durham | 45% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Hedworth | Durham | 27% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Jarrow | Durham | 78% | M: Heriots | R. Lomas, 129 |
| Middlestone | Durham | 70% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Monk Hesleden | Durham | 44% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Monkton | Durham | 21% | M: Heriots | R. Lomas, 129 |
| Monkwearmouth | Durham | 67% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Moorsley | Durham | 45% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Nether Hedworth | Durham | 72% | M: Heriots | R. Lomas, 129 |
| Newton Bewley | Durham | 48% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
| Newton Ketton | Durham | 42% | M: Heriots | Benedictow, Black Death, 367 (citing R. Lomas) |
Historians have expended enormous effort estimating the mortality rates of the Black Death in 1348–1349. English historians have, in fact, probably done more to advance data collection of these rates than scholars elsewhere because of the variety and quality of documentary material that survives for England, particularly for peasants, who comprised over 80 percent of the total population.[39] Although estimates of mortality rates calculated for well-off groups such as wealthy tenants-in-chief of the king and beneficed clergy are useful, mortality assessments for the rural population bring us closer to how many people the Black Death actually killed.[40] Such estimates for the rural population are not unproblematic given the innovative methodologies that scholars have imposed on recalcitrant sources to extract mortality rates. One method involves counting the number of heriots or death-duties paid by peasants on a manor during a specified period, and comparing the number with the total manorial tenants liable to heriot, an exercise that has produced death rates for many different settlements across England (Table 1).[41] Another method relies on a customary head-tax due from a particular class of manorial tenants. On the manors of Glastonbury Abbey, landless men who represented the poorest sector of village society (called garciones and over the age of 12) had to make an annual payment (called chevagium, or head tax), which allows mortality rates to be calculated by subtracting the total for the plague year from the total for the previous year (Table 1).[42] A similar tax was the tithing penny due on some Essex manors from all male villeins (serfs) over age 12; the total sums collected, at the rate of one or one-half penny per head, survive for many years, thus theoretically allowing changes in the size of a village’s population to be determined.[43] Unfortunately, few Essex tithing lists survive for all the years of the Black Death, so the mortality rates are calculated as the percentage decline in those paying tithing penny between a three- to five-year period.[44] Detailed analyses of manors with especially good court rolls and account rolls have also allowed historians to calculate plague mortality rates of at least 40 percent in Halesowen and over 50 percent on the small but heavily populated manor of Hakeford Hall in the Norfolk village of Coltishall.[45]
The mortality rates derived from these different methods show variations ranging from 20 to 100 percent (Table 1), but the overall average mortality of adult males in the countryside was about 50 percent. Because landless males had a generally higher mortality rate of 56–57 percent according to the drop in annual payments made by the poor garciones on Glastonbury Abbey estates (distributed over four counties), and because mortality rates from heriots exclude women, children, and landless men, there have also been attempts to push this overall figure above 60 percent to account for the many poorer peasants and women left out of the heriot and tithing-penny figures.[46] Whether the figure is 40, 50, or even higher than 60 percent, the Black Death of 1348–1349 had an extremely high mortality, in stark contrast to modern plague mortality, which is rarely more than 2–3 percent even in the absence of antibiotic treatment.[47] During the late 1890s and early 1900s, bubonic plague killed approximately one million people per year in India.[48] Though this is a disturbingly high number of people, it does not approach the high levels of mortality that occurred during the Black Death in terms of the proportion of the population killed.
Questions about why the Black Death of 1348–1349 was so deadly remain unanswered by both historians and scientists, although there is some hope that bioarchaeology and paleomicrobiology may provide solutions one day.[49] We now have clear evidence that the Black Death was caused by Y. pestis and in particular by a strain of the bacterium ancestral to most extant strains. Comparison of Y. pestisDNA from skeletons of individuals who died during the Black Death with modern references has so far failed to reveal any significant functional differences in sections of the genome associated with virulence.[50] Comparisons between Y. pestis DNA from different historical epidemics suggest that the First (the Plague of Justinian in the sixth century) and Second (the Black Death) Pandemics represented independent emergences of plague from animals into humans and that the strain responsible for the Plague of Justinian might have subsequently gone extinct.[51] It is also noteworthy that a recent successful analysis of a tooth sample from a Black Death victim, which identified dozens of pathogenic, environmental, and microbiomic taxa, indicates that we have the capability to examine the diversity of pathogens that were circulating at the time of the Black Death.[52] We can thus potentially test the hypothesis that co-infection with multiple pathogens, and resulting within-host competition, drove mortality during the Black Death to extreme levels. If molecular studies of Black Death remains do not reveal any features of the pathogen (or pathogens) that can fully explain the epidemiology of the epidemic, we must look carefully at the characteristics of the human populations that first encountered these pathogens in 1348 to understand the behavior of the fourteenth-century disease.
Recent research suggests that health in general declined before the Black Death emerged in the fourteenth century, which might have contributed to the extraordinarily high mortality of the epidemic.[53] Bioarchaeological analysis of human skeletons dating from A.D. 1000 to 1250 reveals declining life expectancies in London across this period, during which there were repeated famines in England resulting from climatic changes.[54] Since longevity (life expectancy) is a commonly used measure of a population’s general health,[55] these demographic trends estimated from the skeletal data suggest a decline in health prior to the Black Death that might have exacerbated vulnerabilities to a new disease and rendered the epidemic more deadly than it would have been had it struck a more resilient population. These studies have not yet included skeletal samples from late thirteenth century through the mid-fourteenth century, so it remains to be seen whether these declines in health were sustained right before the Black Death.
Who the Black Death Killed: Wealth, Status, Sex, Locale, and Age
Together historical and bioarchaeological sources can offer a great deal about differential mortality, that is, how wealth, position, age, sex, or frail health might have influenced an individual’s susceptibility to plague. These data not only improve our understanding of how the Black Death shaped demographic, economic, political, and social conditions of post-plague medieval populations, but they can also clarify our understanding of the dynamics of emerging infectious diseases today, particularly how the human context can shape the epidemiology and evolution of these diseases.
Contemporary documents tell us precious little about who was infected by the Black Death, although most medieval chroniclers claimed that the Black Death of 1348–1349 was indiscriminate in attacking those of all conditions, age, and sex. Only a few contemporaries noted contrary sentiments, such as: “few, virtually none, of the lords and great men died in this pestilence.”[56] Other documentary sources, such as royal genealogies and inquisitions post mortem for well-off tenants-in-chief of the crown, do show that there were very few casualties among the English royal family, that the higher nobility had a mortality rate of perhaps 4.5 percent in 1348 and 13 percent in 1349, and that the mortality rate among wealthy land-owners may have been a relatively low 27 percent.[57] English bishops also had a better chance of survival (fewer than 18 percent died in 1348–1349) than the secular and monastic clergy, about 45 percent of whom died in the Black Death.[58] The clerical mortality rates were on average lower than those of the rural majority. Wealth may have mattered since the poorest sector of rural society, those who could neither lease nor own land (marked as “LL,” “landless men” in Table 1), died in proportionately greater numbers than more well-off peasants.
Until recently, neither historians nor bioarchaeologists could offer firm evidence that the Black Death of 1348–1349 hit one sex harder than the other, though some medieval chroniclers of later plagues, such as that of 1360–1361, believe the plague killed more men.[59] The documentary evidence relates largely to men, although one study of rural manors in the northern county of Durham found that 52 percent of female tenants died in the Black Death compared to 50 percent of the total tenant population[60] More convincing evidence of greater female mortality during outbreaks of late medieval plague, including the Black Death, has been found in a recent analysis of thousands of death duties (called mortmains) paid in the Hainaut region of the southern Netherlands. Mortmain payments differed from heriots in taxing not the landholding of a tenant, but individuals, including women and young adults who were not yet heads of households. The study found an overall average sex ratio for mortality of 107 in the period 1349–1450, but 94 in plague years and 89 during the Black Death. Further tests on these data suggest that women were disproportionately affected by plague because they biologically had less resistance to the disease than men, rather than because of their differential exposure given their greater numbers in towns or the time they spent at home as caregivers.[61] This exciting new evidence will hopefully stimulate more historical and bioarchaeological studies to help resolve the conflicting evidence about sex-selective mortality during periods of plague.
Even less certainty surrounds the extent to which the Black Death was more virulent in urban than rural areas. One leading historian of plague maintains that rural regions suffered greater mortality than towns, but most historians argue the reverse based on solid data showing high mortality in pre-modern towns and their reliance on migration to maintain and grow their populations.[62] Unfortunately, there is no systematic source of documentary data to measure urban plague mortality such as we have for the clergy, wealthy landowners, and rural dwellers. The urban evidence tends to be indirect and peculiar to individual towns. A comparison of tax returns from Canterbury, for instance, shows that two-thirds of the tax-paying population vanished between 1346/7 and 1351/2, while monastic records show that one-half of the tenants of Rochester Priory in Rochester died and two-thirds of those residing at Durham Priory in Durham.[63] Wills have also been exploited to study trends in urban mortality. A careful study of wills in medieval London, for example, shows that the mortality rate in 1349 was seventeen times higher than in other years, which translates into a mortality rate of about 51 percent if we accept the average of a 3 percent annual mortality rate calculated for early modern populations.[64] It is likely that the actual mortality rate in London was higher, given the relative wealth of will-makers.
Finally, even more debatable is whether age made any difference to susceptibility to plague. Chroniclers occasionally declared that plague targeted the young or old or those in the prime of life, but age was rarely mentioned in accounts of the first wave of the plague in 1348–1349.[65] There is some questionable evidence that older men faced higher risks of mortality compared to younger men[66] (hardly surprising given their greater frailty), which bioarchaeological research has shown did make individuals more susceptible to the plague.
Bioarchaeology can provide especially useful insights into selective plague mortality because it is possible to determine the sex, age, and general health of skeletal remains, even though it cannot establish whether an individual died from plague without using molecular methods, most of which are expensive and destructive. A good deal of this work is statistical and involves the application of methodologies designed to compensate for the limitations of bioarchaeological data. As with historical documents, not every person who lived in the past is included in the observable bioarchaeological record. Certain individuals might be excluded from the samples analyzed because they were buried in unexcavated areas or because their bones were badly preserved, a particular problem for the smaller and thinner bones of infants, who are very often under-enumerated in skeletal samples.[67]
Partial excavation of a site, which is a common result of time and financial constraints, can produce biased skeletal samples if burial locations within the cemetery were influenced by factors like age, sex, socioeconomic standing, or disease status. Another problem relates to the traditional methods of aging skeletons, which tend to underestimate older adult ages,[68] or to group them into broad age intervals, such as “50+ years.”[69] These methods are not capable of making distinctions between, for example, a 50- and a 90-year-old, two ages with very different morbidity and mortality regimes. Compounding the problems with age estimation is the standard use of mean age-at-death or life tables to infer mortality patterns, both of which implicitly assume the population under consideration was stationary: that is, closed to migration, with zero population growth, and unchanging age-specific mortality and fertility rates.[70] Departures from these assumptions—which occur, for example, if the population was growing or contracting—means that skeletal age patterns might not accurately represent past trends in mortality patterns.[71]
Human bones often contain specific signs (such as lesions, pitting, thickening, and cracks) that reflect exposure to physiological stress in general or particular health problems, such as malnutrition, tuberculosis, previously broken bones, and other conditions. These health problems can shed light on selective mortality in terms of what might have made certain individuals more susceptible to death from plague. This type of analysis cannot be taken too far, however, because not all of the genetic, physiological, social, and environmental variations affecting frailty (that is, susceptibility to disease and death) leave discernable signs on the skeleton.[72] Reconstructing health from skeletons is further complicated because skeletal lesions, which form in response to disease, malnutrition, or trauma, might not always indicate poor health. Lesions could actually point to relatively good health because they reflect the ability of individuals to survive the associated stressor long enough for the lesions to form. Similarly, the absence of a lesion might suggest relatively poor health if individuals without them were so frail that they quickly succumbed to stressors and died before lesions had a chance to form.[73] In the absence of other supporting evidence, we thus cannot interpret the presence or absence of skeletal lesions in a straightforward manner, a problem articulated as the Osteological Paradox.[74]
Many of the published bioarchaeological analyses of the fourteenth-century plague have been done using skeletons buried in the East Smithfield cemetery in London.[75] The documentary history of the East Smithfield cemetery indicates that it was used only during the Black Death, so most if not all of the over 600 individuals recovered from the cemetery were victims of the 1348–1349 epidemic.[76] Sharon DeWitte previously compared East Smithfield to a non-epidemic, pre–Black Death assemblage to assess whether risks of death during the Black Death varied by age, sex, or pre-existing health condition (frailty). The results indicate that elderly adults faced higher risks of death compared to younger adults during the Black Death, as occurs under normal mortality conditions.[77] The mortality pattern for children in East Smithfield is less clear, however, perhaps as a result of the small sample of infants and children available from the cemetery.[78] Assessment of sex differences in mortality during the Black Death failed to reveal a significant difference in risk between men and women during the epidemic.[79] Finally, DeWitte and colleagues found that the Black Death disproportionally killed people with skeletal lesions compared to those without them.[80] These results suggest that people who experienced physiological stressors before the Black Death were subsequently more likely to die during the epidemic compared to their peers who lacked the lesions, indicating that the epidemic was selective with respect to frailty.
In summary, the combined evidence from the historical and bioarchaeological sources indicates that wealth, status, health and perhaps age affected risks of death during the Black Death. The elderly, those who were impoverished, and those who had suffered relatively poor health faced higher risks of death during the epidemic than their younger, wealthier, and healthier peers. Neither source of information for England has yielded substantial evidence that one sex fared better than the other, although recent work on the southern Netherlands offers strong evidence that plague more negatively affected women than men. Further documentary work on towns would be especially welcome to address the extent to which urban mortality might have exceeded rural mortality; this question is more difficult to answer through bioarchaeology since plague cannot be diagnosed from skeletal remains without expensive DNA analysis and since the population at risk cannot be determined in cemeteries. Bioarchaeology does, however, have the potential in the future to clarify the risk of mortality for the youngest individuals in the population during the Black Death once additional Black Death cemeteries are excavated, thereby increasing the available sample sizes for analysis. Isotopic analysis of carbon and nitrogen will also be able to reveal more clearly how diet or nutritional stress, two factors that can produce skeletal lesions, affected risks of death during historical plague epidemics.
Who the Black Death Killed in 1361–1362
In contrast to their remarks that the Black Death of 1348–1349 killed indiscriminately, chroniclers were far more emphatic in describing the age- and sex-specific mortality of the plague of 1361–1362, a topic that has received relatively little attention from historians and bioarchaeologists alike. This “second pestilence,” differed first of all in targeting the young. Medieval English chroniclers called it “the mortality of children” or commented on the “death of children and adolescents,” or that “children and adolescents were the first to die, and then the elderly,” and they claimed that “both rich and poor died, but especially young people and children.” Secondly, the chroniclers emphasized the second pestilence’s effect on men, saying that it “devoured men rather than women”; noting, for example, that “for this year the mortality was particularly of males.” Several spoke of both age and gender, labeling it “a mortality of men, especially of adolescents and boys, and as a result it was commonly called the pestilence of boys.”[81] This visitation of the plague also seems to have focused more on southern England, and was said to have started in London where it was so devastating in May and June of 1361 that the royal courts had to be adjourned, a timing confirmed by the peak in wills during the summer.[82] This was the plague referred to in the well-known graffiti inscription in the Hertfordshire church of Ashwell: “wretched, fierce, violent . . . [only] the dregs of the populace live to tell the tale.”[83] Although the 1361–1362 plague was the second deadliest in medieval England, we know far less about it in part because the chroniclers had less to say about it, but mostly because historians have not fully exploited what sources are available.
Analysis of the institutions to benefices for the better-off secular clergy shows a mortality rate of about 10–30 percent in 1361–1362, compared to their 45 percent average in 1348–1349.[84] In 1361–1362, evidence from heriots show that the mortality rate on the Hampshire manor of Bishop’s Waltham was 13 percent and 12–18 percent on the Dorset manor of Titchfield, while court roll references to deaths at the manor of Halesowen indicate a mortality rate of a little over 9 percent.[85] The evidence from wills in London suggests a mortality rate of almost 20 percent.[86] This range of mortality rates in the “second pestilence” appear insignificant when compared to the death rates in 1348–1349, but they were higher than for any subsequent visitation of the plague.[87] There is less evidence that wealth—and the better diet, health, and sanitary conditions associated with it—had the impact in the 1361–1362 plague that it may have had in the earlier plague. Although references to the lower mortality of the rich are not particularly prominent among observers of the 1361–1362 plague, inquisitions post mortem suggest that the morality rates of the nobility and tenants-in-chief were relatively higher than in 1348–1349 and closer to the mortality levels suffered by the general population.[88]
We have some suggestive evidence about the sexual bias of the 1361–1362 plague in a study of entry fines paid by new villein tenants on the estate of the bishop of Winchester, which included almost sixty manors located mainly in Hampshire, but also in the West Midlands and West Country.[89] The fines provide a good record of all post-mortem property transactions, while also offering enough evidence on the parties involved to establish the relationship between the deceased and the individual who took over the property. The fines reveal a rise in the numbers and proportions of women inheriting in 1361–1362 compared to previous years, and a marked downturn in the percentage of sons and other male kin who inherited, suggesting greater mortality for men, since women inherited only in the absence of male heirs. It might also be telling that the percentage of widows who inherited rose the most, while potentially younger women (such as sisters, daughters, nieces, and granddaughters) did not inherit any more than they had in earlier decades, implying that older women may have better survived the second plague.
Bioarchaeological analysis reveals some different answers to the question of who was killed by the second wave of the Black Death in 1361–1362. For this essay, DeWitte analyzed data from burials associated with the Cistercian Abbey of St. Mary Graces in London, which overlies the earlier East Smithfield cemetery. The St. Mary Graces assemblage includes burials within the Abbey church and chapels and a larger lay cemetery; within the cemetery, there are nearly 200 burials attributed to the 1361 plague.[90]
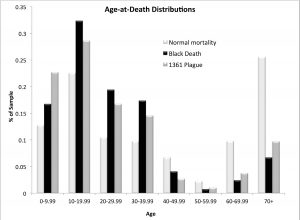
Figure 2: Age-at-death distributions from the East Smithfield Black Death (1348-9) cemetery, and St. Mary Graces plague of 1361-2 and non-plague (normal mortality) burials. Produced by Sharon DeWitte.
| Age at death | Non-plague | Black Death | 1361-1362 plague | |||
|---|---|---|---|---|---|---|
| 0–9.99 | 17 (12.78%) | 82 (16.8%) | 42 (22.7%) | |||
| 10–19.99 | 30 (22.56%) | 158 (32.3%) | 53 (28.6%) | |||
| 20–29.99 | 14 (10.53%) | 95 (19.4%) | 31 (16.8%) | |||
| 30–39.99 | 13 (9.77%) | 85 (17.4%) | 27 (14.6%) | |||
| 40–49.99 | 9 (6.77%) | 20 (4.1%) | 5 (2.7%) | |||
| 50–59.99 | 3 (2.26%) | 4 (0.8%) | 2 (1.1%) | |||
| 60–69.99 | 13 (9.77%) | 12 (2.5%) | 7 (3.8%) | |||
| 70+ | 34 (25.56%) | 33 (6.7%) | 18 (9.7%) | |||
| Total | 133 | 489 | 185 |
Table 2: Age-at-death distributions from the East Smithfield Black Death cemetery and the St. Mary Graces cemetery (1361–1362 plague and non-plague). Percentages refer to proportion of total sample.
DeWitte assessed mortality patterns in the second outbreak of plague, specifically whether it was selective with respect to sex, age, and frailty. The age-at-death distributions (Table 2) from the two sets of plague burials in 1348–1349 (East Smithfield cemetery) and in 1361–1362 (St. Mary Graces) do not differ significantly from each other, but each is different from the normal mortality (non-plague) distribution from St. Mary Graces.[91] There is some evidence that children and people in the prime of life were more likely to die during outbreaks of plague since the plague burials had more people below the age of 40 and fewer adults above the age of 70 than the non-plague burials in St. Mary Graces. The non-plague burials might be biased toward older individuals, however, since they include individuals who were buried within the Abbey of St. Mary Graces itself, such as monks and wealthy lay people. Of note, there is a higher proportion of children (under age 10) in the 1361 plague burials compared to both the 1348–1349 and the non-plague burials (Figure 2). The 1361–1362 burials contain 10 percent more children than the non-plague burials, and nearly 6 percent more children compared to the earlier Black Death burials (though the latter difference is not statistically significant).
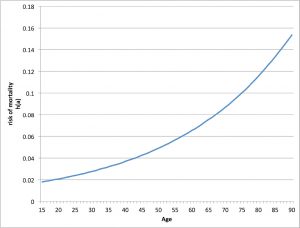
Figure 3: Hazard of adult mortality for the 1361-2 plague burials in St. Mary Graces cemetery. Produced by Sharon DeWitte.
The higher proportion of children in the 1361–1362 plague burials is consistent with the descriptor “mortality of children” for the second outbreak of plague, which could reflect age-related patterns of vulnerability following the Black Death.[92] Individuals who survived the Black Death (and were above the age of 10 during the second wave) might have acquired immunity to resist the disease whereas those born since the epidemic lacked acquired immunity and thus were susceptible to the disease.[93] The (statistically) non-significant difference in age distributions between the two plague burials might mean that the second outbreak was not more lethal to children than the first. These results, however, could also be an artifact of relatively small sample sizes or the poor preservation of infants and young children that affects skeletal assemblages in general. To further examine mortality patterns, the age-at-death data from the 1361 plague burials were used to estimate the parameters of a model of the age pattern of adult mortality.[94] The results indicate that the risk of death increased with adult age during the second outbreak of plague (Figure 3), as was previously observed for the Black Death and for conditions of non-epidemic mortality.[95]
Though there are more adult males than adult females in the 1361–1362 burials (52.7 percent vs. 47.3 percent), as is true for the 1348–1349 and non-plague burials, results from an assessment of differences in risk of mortality between males and females during the 1361 plague do not corroborate the historical evidence that men may have been more susceptible to the 1361–1362 plague.[96] The 95 percent confidence interval for the estimated effect of sex on adult mortality includes both positive and negative values, which indicates a lack of significant difference in risk of death during the 1361 plague between men and women, as was earlier reported for the Black Death and pre–Black Death normal mortality samples. It is possible that (as noted for previous analyses of East Smithfield), this result might be an artifact of the way in which the effect of sex was modeled. Nonetheless, this result raises the question of how we might resolve discrepancies between historical and bioarchaeological evidence. The most conservative approach is to, at least temporarily, assign greater weight to the positive evidence (provided in this case by the historical evidence) and allow the tension between the two lines of evidence to stimulate further analysis in the event that additional data become available.
There was, however, good evidence that the 1361 outbreak of plague was selective for frailty as was the case during the Black Death.[97] For this essay, DeWitte focused on the risk of death associated with the presence of periosteal new bone formation, a lesion that can form in response to stimuli that traumatize the tissue that covers the surfaces of bone, which in turn results from local or systemic infection or inflammation associated with a variety of factors.[98] The analysis suggests an elevated risk of mortality for those with the skeletal lesion among the 1361 plague victims, as has been found for normal mortality samples and among Black Death victims.[99] In summary, the bioarchaeological evidence indicates that age and frailty, but not sex affected risks of mortality during the second wave of plague. These results demonstrate the ability of bioarchaeological data to confirm or refute historical evidence as well as expand the lines of inquiry beyond those possible with existing historical evidence.
Conclusion
Scholars of medieval plague have only recently combined bioarchaeological, historical, and paleomicrobiological information to achieve a fuller understanding of the epidemiology of the disease and its relationship to modern plague. Until a few years ago when Y. pestis was definitively identified as the bacterium responsible for both medieval and modern plague, efforts to compare historical and scientific data on the symptoms, pace, duration, and mortality of plague tended to generate huge controversies in which some scholars adamantly claimed the Black Death was not bubonic plague while others just as ardently argued the opposite. New discoveries in bioarchaeology and paleomicrobiology have in particular moved these debates toward a resolution, and are opening up more fruitful avenues regarding how health, age, social status, wealth, and sex may or may not have affected chances of survival.
Chronicles, along with data drawn from institutions to benefices for the secular clergy, inquisitions post mortem for well-off landowners, wills, and, most importantly, manorial sources about the rural majority reveal much about the temporal and spatial spread of the Black Death, as well as mortality rates, especially the far higher death rates in the 1348–1349 plague compared to the second pestilence in 1361–1362. These historical sources also point to how wealth and social status influenced risks of mortality during the Black Death, and how age and sex might have affected risks during the plague of 1361. The bioarchaeological evidence uniquely reveals that health status affected risks of death during both outbreaks, which historical evidence simply cannot reveal in any systematic way. By combining the historical and bioarchaeological data, it becomes clear that risks of mortality during at least the first two outbreaks of medieval plague varied along several dimensions including social status, wealth, age, and health status.
Neither bioarchaeological data nor historical evidence provides a flawless perspective on patterns of mortality during medieval plague epidemics, or more generally of life, health, and death in the past. Historical data from the medieval period tend to be biased toward wealthy men, blinding us to the experiences of the majority of the population that was at risk of disease and death during the Black Death. Medieval chroniclers and other record keepers did not take note of many variables modern epidemiologists consider essential to understanding disease processes, so we do not have access to details about the age, sex, and health status of medieval plague victims and survivors from written records.
Cemeteries, on the other hand, tend to be more inclusive than written records, yielding bioarchaeological data on women, children, and the poor, and thus allowing for reconstructions of health and death for a broader sample of once-living populations. Bioarchaeological samples are, however, subject to their own sources of bias, such as differences in preservation, inaccuracies in age estimation, and incomplete excavation. Further, there are relatively few burials clearly associated with medieval plague epidemics, particularly from rural areas, and social status is largely invisible in the burials that do exist.
Ideally, an interdisciplinary approach to Black Death epidemiology allows the strengths of each line of evidence to compensate for the weaknesses in the other. The combination of the bioarchaeological and historical evidence, when possible, can provide insights that are not possible when each is analyzed in isolation. As shown here with respect to sex differences in plague mortality, this approach might highlight discrepancies in the patterns revealed by the two lines of evidence. Such tensions will ideally motivate further study.
Notes
-
Campbell, Great Transition, 289–313; Green, “Editor’s Introduction,” and the essays in Green, ed., Pandemic Disease.
-
M. Ziegler, “Black Death,” 260; Little, ed., Plague and the End of Antiquity; Perry and Fetherston, “Yersinia pestis,” 36.
-
M. Ziegler, “Black Death, 260.
-
Those who argued that the Black Death was not bubonic plague include Twigg, Black Death; Scott and Duncan, Biology of Plagues; Cohn, Black Death Transformed. For the recent discoveries, see Drancourt et al., “Detection”; Raoult et al., “Molecular Identification of Yersinia pestis.” These early results were viewed with skepticism because of the use of small, poorly dated samples and because of the possibility of contamination by modern Y. pestis; see Twigg, “Black Death”; Wood and DeWitte-Aviña, “Was the Black Death Yersinial Plague?”; Gilbert et al., “Absence,” 348, 352. More recent DNA and immunological studies have yielded more convincing evidence: Bianucci et al., “Rapid Diagnostic Test”; Bos et al., “A Draft Genome”; Haensch et al., “Distinct Clones”; Kacki et al., “Black Death”; Pusch et al., “Yersinial F1 Antigen”; Schuenemann et al., “Targeted Enrichment”; Tran et al., “Co-Detection.” In 2011, the entire genome of fourteenth-century Y. pestis was reconstructed from DNA fragments isolated from the East Smithfield Black Death burial ground in London; Bos et al., “A Draft Genome.” For the East Smithfield cemetery, see also below.
-
Perry, “Is Bioarchaeology a Handmaiden?” 489.
-
For contemporary descriptions, see Horrox, Black Death, 14–92. See also the descriptions of symptoms culled from an impressive array of medieval sources in Cohn, Black Death Transformed, 57–139, although marshalled to argue that the Black Death was not modern bubonic plague.
-
Boccaccio, Decameron, 50–51.
-
Horrox, Black Death, 81.
-
Chroniclers often noted the days victims were ill before dying, as in Horrox, Black Death, 19 (three days), 25 (three to five days), 27 (three days, some more rapid), 35 (one to three days), 36 (three days), 42 (two days), 55 (two to three days), 64 (three to four days), 74 (three days), 77 (two to three days), 84 (two days).
-
Perry and Fetherston, “Yersinia pestis,” 57–58; M. Ziegler, “Black Death,” 266.
-
Some (Cohn, Black Death Transformed,64–65, 68–69, 77–81) argue that buboes or boils arose in the neck and armpits in medieval plague and more often in the groin in modern plague.
-
Higgins, “Emerging or Re-Emerging,” 576–77; Cleri et al., “Plague Pneumonia,” 12. Pharyngeal and gastrointestinal plague are caused by ingestion of contaminated meat; M. Ziegler, “Black Death,” 266.
-
Perry and Fetherston, “Yersinia pestis,” 58; Lathem et al., “Progression,” 17787.
-
Horrox, Black Death, 4–5, 43, 63.
-
For critical (and tendentious) discussions of this view, see Benedictow, Black Death, 27–31, 236–41; Benedictow, What Disease Was Plague? 491–92. Person-to-person transmission occurs when infected individuals cough droplets containing the plague bacteria into the air, which are then inhaled by others in close proximity (see Begier et al., “Pneumonic Plague Cluster,” 464; Hinckley et al., “Transmission Dynamics,” 554; Pechous et al., “Pneumonic Plague,” 191.
-
For example, see Richard et al., “Pneumonic Plague.”
-
Hinckley et al., “Transmission Dynamics.”
-
Nishiura, “Epidemiology,” 1061.
-
P. Ziegler, Black Death, 122–27; Benedictow, Black Death, 126–29.
-
Horrox, Black Death, 64–65.
-
Ibid., one year: 62, 63; more than a year: 81; two years: 64.
-
The other main outbreaks of plague in late fourteenth-century England were in 1361–1362, 1369, 1374–1379, and 1390–1393; their exact dates are open to question since they seem to have become increasingly regional. See ibid., 85–92; Shrewsbury, Bubonic Plague, 126–56 (although he asserts these pestilences were not bubonic plague).
-
Previous studies are discussed in Benedictow, Black Death, 123–26, 343–59.
-
Wood, Ferrell, and DeWitte-Aviña, “Temporal Dynamics”; Benedictow, Black Death, 123–26, 342–48.
-
For example, Lock, “Black Death.”
-
For examples, see Megson, “Morality”; Röhrkasten, “Trends of Mortality,” 184–89; Sloane, Black Death in London, 43–86.
-
Lewis (“Disaster Recovery”) assessed thousands of datable potsherds from test-pit excavations from nearly 2000 archaeological sites in eastern England. There was a 44.7 percent decrease overall (p. 780) in the number of pits yielding potsherds following the Black Death, which may reflect declines in the size of the human population. This approach allows for the mapping of both the occurrence and severity of population decline associated with the epidemic.
-
For example, Sloane, Black Death in London; Watts, “Black Death”; James, Black Death, and the map in Gummer, Scouring Angel, 422 (reproduced as Figure 1, below).
-
Benedictow, What Disease Was Plague? 420–84. For modern plague, Bacaër, “Model of Kermack,” 410; Kreppel et al., “A Non-Stationary Relationship,” 3; Migliani et al., “Epidemiological Trends,” 1232; Moore et al., “Seasonal Fluctuations,” 2.
-
Benedictow, Black Death, 124–42; the figures are on p. 142. The spread of both medieval and modern plague is discussed in Benedictow, What Disease Was Plague?, 151–93, albeit with the aim of defending his previously published views.
-
Benedict, Bubonic Plague, 108.
-
Horrox, Black Death, 44, 81, 107–08; quotation at 83.
-
Hinnebusch, “Bubonic Plague,” 646.
-
M. Ziegler, “Black Death,” 266; Hinnebusch, “Bubonic Plague,” 646; Perry and Fetherston, “Yersina pestis,” 37, 51–53.
-
Royer, “Blind Men,” 108.
-
Drali et al., “A New Clade”; Eisen et al., “Early-phase Transmission”; Houhamdi, et al., “Experimental Model”; Ratovonjato et al., “Yersinia pestis”; M. Ziegler, “Case for Louse-Transmitted Plague.”
-
Mathematical models reduce complex phenomena to a manageable number of variables and the relationships between them. Infectious disease models can be used to predict the behavior of diseases or can be compared to observations of past diseases in an attempt to understand transmission patterns, mortality rates, or other epidemiological variables and their outcomes. Dean (“Modeling Plague Transmission”) used an SIR (Susceptible-Infected-Recovered) model to assess the spread of the Black Death assuming different vectors, including the louse.
-
Royer, “Blind Men,” 101.
-
For others who agree on the special value of English sources for estimating mortality, see Benedictow, Black Death, 126; Olea and Christakos, “Duration,” 296.
-
See below for estimates of the mortality of the well-off.
-
The method depends on the fortunate survival of manorial account rolls or court rolls (the documents where heriots were liable to be recorded), as well as documentation (such as a rental) showing the total number of customary tenants on the manor; see Postan and Titow, “Heriots and Prices,” and Titow, English Rural Society, 69–70. See also Ohlin, “No Safety,” 85–89, for further criticisms of the method.
-
Ecclestone, “Mortality”; Titow, English Rural Society, 71.
-
Tithings were part of the frankpledge system. Russell, British Medieval Population, 226–27; Poos, A Rural Society, 106–07. See also Postles, “Demographic Change.”
-
The numbers taxed, moreover, could have changed due to in- or out-migration, or lack of administrative fervor during the plague in enforcing the tax, all of which further reduce the tithing lists’ usefulness as an indicator of population losses through plague mortality. Poos, A Rural Society, 106–08, recognizes these problems. See also Benedictow, Black Death, 369–74, for a detailed critique, although his conclusion (p. 373) that the average of the tithing mortality figures of c. 43–46 percent need to be adjusted upward to 60 percent is based on educated speculation, not firm numbers.
-
Razi, Life, Marriage and Death, 106–07; Campbell, “Population Pressure,” 96.
-
Arthur, “Black Death”; Benedictow, Black Death, 376–77. Benedictow’s assumptions about the “super-mortality” of women, are, however, based on mortality rates in modern not medieval plague; see Kowaleski, “Gendering,” 185.
-
His Majesty’s Stationary Office, “Statistical Abstract,” 232–35.
-
Perry and Fetherston, “Yersina pestis,” 36.
-
Paleomicrobiology is the study of ancient biomolecules such as DNA recovered from human and other animal remains.
-
Bos et al., “A Draft Genome,” 509.
-
Wagner et al., “Yersinia pestis”; Wiechmann and Grupe, “Detection.”
-
Devault et al., “Ancient Pathogen DNA.”
-
DeWitte, “Setting the Stage. “
-
Ibid.
-
Gage, “Are Modern Environments Really Bad for Us?” 97.
-
Horrox, Black Death, 63, 64.
-
Ibid., 35; Russell, British Medieval Population, 216; P. Ziegler, Black Death, 227–30; McFarlane, Nobility, 169–70.
-
For bishops, see Coulton, Medieval Panorama, 496, 749. For monastic clergy, see Russell, British Medieval Population, 223 (45 percent based on a small sample of 566 monks in twelve monasteries); he also calculated a 41.5 percent mortality rate for abbots and priors who died in 1349–1350 (p. 225). For an extended discussion of how to interpret the mortality figures for secular clergy based on institutions to benefices, see Benedictow, Black Death, 343–59.
-
For the conflicting evidence on sex and plague mortality, see Kowaleski, “Gendering,” 185. For recent medieval and early modern opinions, see Curtis and Roosen, “Sex-selective Impact.” For bioarchaeological evidence on sex, see below, and for the 1360–1361 plague, see the next section, below.
-
R. Lomas, “Black Death,” 130 (based on 155 female tenants of a total of 718 tenants at risk). When women are subtracted from the total, the mortality rates of women and men were virtually the same.
-
Curtis and Roosen, “Sex-selective Impact.”
-
Benedictow, Black Death, 260–61; id., “Black Death.” For a discussion of the overall greater mortality and morbidity in towns, see Kowaleski, “Medieval People,” 583–93.
-
Butcher, “English Urban Society,” 93–97; Britnell, “Black Death in Durham,” 47. The best overview is in Britnell, “Black Death in English Towns.”
-
Röhrkasten, “Trends of Mortality,” 182.
-
Horrox, Black Death, 62–85; only one chronicler of the 1348–1349 plague noted age in claiming that more “young and strong” people died (p. 81). For 1361–1362, see below, note 79.
-
Russell, British Medieval Population, 216–18; Russell’s figures, however, are highly questionable since he did not allow for “normal” deaths amongst an older population; see Ohlin, “No Safety,” 78–80, and Poos, Oeppen, and Smith, “Re-assessing.” Razi (Life, Marriage and Death, 107–09) presents data showing that male peasants in their 50s in the parish of Halesowen had a higher mortality rate than younger men, but admits how crude his roundabout method of calculation is, and how small the numbers are.
-
Lewis, Bioarchaeology of Children, 30–33; Milner, Wood, and Boldsen, “Paleodemography,” 573–74.
-
This occurs because these methods are biased toward known-age reference samples (a problem called age-mimicry), which tend to be composed predominantly of young individuals; see Bocquet-Appel and Masset, “Farewell to Paleodemography”; Buikstra and Konigsberg, “Paleodemography”; Müller, Love, and Hoppa, “Semiparametric Method”; Van Gerven and Armelagos, “Farewell to Paleodemography?”; Milner and Boldsen, “Transition Analysis.”
-
Buikstra and Konigsberg, “Paleodemography”; Boldsen et al., “Transition Analysis.”

-
Milner, Wood, and Boldsen, “Paleodemography,” 563; Wood et al., “Osteological Paradox,” 344.
-
In some cases, the distribution of skeletons’ ages might, counter-intuitively, be more reflective of fertility than mortality; Sattenspiel and Harpending, “Stable Populations.”
-
Wood et al., “Osteological Paradox,”345; Vaupel, Manton, and Stallard, “Impact of Heterogeneity,”439.
-
Ortner, “Theoretical and Methodological Issues,” 9–10; Wood et al., “Osteological Paradox,” 353–54; DeWitte and Stojanowski, “Osteological Paradox,” 407–08.
-
Wood et al., “Osteological Paradox.”
-
Grainger and Hawkins, “Excavations at the Royal Mint Site”; Grainger et al., The Black Death Cemetery; Hawkins, “Black Death,” 637–38. There are a limited number of excavated burial grounds associated with the Black Death or subsequent outbreaks of plague. In addition to East Smithfield, in London there are burials in the St. Mary Graces cemetery attributed to the second outbreak of plague in 1361 (detailed below). There are also known plague burials from the Black Death or afterward from Hereford, England (Gowland and Chamberlain, “Detecting Plague”); Germany (Seifert et al., “Genotyping Yersinia pestis”); Barcelona, Spain; Dendermonde, Belgium; France (Castex and Kacki, “Demographic Patterns”); and from Bologna, Italy (Rubini et al., “Mortality Risk Factors”).
-
The Museum of London’s Department of Greater London Archaeology excavated East Smithfield in the 1980s as part of a planned redevelopment project. The collection is currently curated by the Museum of London’s Centre for Human Bioarchaeology.
-
DeWitte, “Age Patterns.” DeWitte used hazards analysis to examine age patterns of mortality. With this approach, a model of mortality (such as the Gompertz model used by DeWitte) is fitted to data from skeletal samples to reveal age patterns of mortality. This approach is suited to small samples, as the mortality models smooth the random variation present in the skeletal data but do not impose any particular patterns on the data. See Wood et al., “Mortality Models”; and Gage, “Bio-Mathematical Approaches.” DeWitte also used the transition analysis method of age estimation; see Boldsen et al., “Transition Analysis.”
-
Only about 8 percent of the East Smithfield sample was below the age of 5, and it is difficult to estimate risks of mortality for children without very large sample sizes. It is not unusual for infants and young children to make up a relatively small proportion of skeletal samples, as mentioned above. Such “infant under-enumeration” is often attributed to the fact that the bones of infants and children are relatively small and thin and thus might be more likely to decay rapidly or be overlooked during excavation compared to the bones of older individuals.
-
DeWitte assessed sex differences by modeling sex as a covariate affecting the Gompertz model of mortality. This approach assumes that sex differences (or lack thereof) in mortality are constant across all ages, which might not be realistic. For example, in many living populations, females face higher risks of death at reproductive ages (because of high maternal mortality resulting from increased susceptibility to some diseases like malaria, complications during childbirth, and other factors) but lower risks at pre- and post-reproductive ages. See DeWitte, “Effect of Sex,” for details. Castex and Kacki (“Demographic Patterns,” 8) also failed to find evidence that the Black Death disproportionately affected either sex using data from Hereford, England and Dreux, France.
-
DeWitte and colleagues used a hazard model (the Usher model) that allows for the estimation of the difference in risk of death between individuals with and without skeletal lesions and that addresses some of the issues associated with the Osteological Paradox. Usher, “A Multistate Model.” See DeWitte and Hughes-Morey, “Stature and Frailty”; DeWitte and Wood, “Selectivity.”
-
Horrox, Black Death, 85–86; Sloane, Black Death in London, 122–23. See also Shrewsbury, Bubonic Plague, 126–32, for further contemporary comments, although Shrewsbury believes it was not bubonic plague.
-
Calendar of Close Rolls 1361–64, 181–82, 197–98; Röhrkasten, “Trends of Mortality,” 192–94; Sloane, Black Death in London, 122–42 (will data at p. 125).
-
Dickens, “Historical Graffiti.” 181.
-
Above, note 58.
-
Titow, English Rural Society, 70; Watts, “Black Death,” 23–25; Razi, Life, Marriage and Death, 103, 127.
-
Mullan, “Mortality, Gender and the Plague of 1361–62,” 9.
-
For the nobility, see McFarlane, Nobility, 169–70. For the data from the inquisitions post mortem of tenants-in-chief, see Russell, British Medieval Population, 217–18, 222, though his calculations are considered somewhat flawed (above, n. 65). See also Nash, “Mortality Pattern.”
-
Mullan, “Mortality, Gender and the Plague of 1361–62,” esp. 22–23.
-
Gilchrist and Sloane, Requiem; Grainger and Phillpotts, Cistercian Abbey; Sloane, Black Death in London, 136–40. It continued until the Reformation in 1538; see Grainger and Hawkins, “Excavations at the Royal Mint Site”; Grainger et al., Black Death Cementery.An associated cemetery was used for burials of the general populations, whereas monks and important lay people were buried within the Abbey’s church and chapels; see also Rogers and Waldron, “DISH and the Monastic Way of Life”; Grainger and Phillpotts, Cistercian Abbey.
-
These results are based on the Kolmogorov-Smirnov test, a nonparametric test that can be used, as in this case, to determine whether two distributions are significantly different from one another.
-
See the discussion of the documentary evidence for 1361–1362, above.
-
A study of long-term recovered plague patients has found antibodies against Y. pestis in people more than ten years following initial infection; see Li et al., “Humoral and Cellular Immune Responses.”
-
This analysis used the same Gompertz model (above, n. 78) that DeWitte employed for studies of East Smithfield. Initial analyses of mortality across the lifespan failed to produce good estimates of the immature component of mortality associated with infant and childhood causes of death.
-
DeWitte, “The Effect of Sex.”
-
The effect of sex (female) on risk of death is 0.23 (95 percent confidence interval: -0.24, 0.64).
-
DeWitte and Wood, “Selectivity.”
-
This analysis was done using the Usher model described above (n. 79, above). For details about periosteal new bone formation, see Larsen, Bioarchaeology, 86–88; Ortner, Identification of Pathological Conditions, 206; Weston, “Investigating.”
-
The effect of having lesions on risk of death is 1.87 (95 percent confidence interval: 1.16, 7.1).
Works Cited
Primary Sources
Boccaccio, G. The Decameron, trans. G. H. McWilliam. Harmondsworth: Penguin Press, 1972.
Calendar of Close Rolls preserved in the Public Record Office, Edward III, vol. 12, 1361–4. London: Her Majesty’s Stationary Office, 1912.
Horrox, R., ed. and trans. The Black Death. Manchester: Manchester University Press, 1994.
https://doi.org/10.7765/MMSO.34985
Secondary Sources
Arthur, P. “The Black Death and Mortality: A Reassessment.” In Fourteenth Century England VI, ed. C. Given-Wilson and N. Saul, 49–72. Woodbridge: Boydell and Brewer, 2010.
Bacaër, N. “The Model of Kermack and Mckendrick for the Plague Epidemic in Bombay and the Type Reproduction Number with Seasonality.” Journal of Mathematical Biology 64 (2012): 403–22. https://doi.org/10.1007/s00285-011-0417-5
Ballard, A. “The Manors of Witney, Brightwell, and Downton.” In Oxford Studies in Social and Legal History, 5, ed. P. Vinagradoff, 181–216. Oxford: Clarendon Press, 1916.
Begier, E. M., G. Asiki, Z. Anywaine, B. Yockey, M. E. Schriefer, P. Aleti, A. Ogden-Odoi, J. E. Staples, C. Sexton, S. W. Bearden, and J. L. Kool. “Pneumonic Plague Cluster, Uganda, 2004.” Emerging Infectious Diseases 12(3) (2006): 460–67.
https://doi.org/10.3201/eid1203.051051
Benedict, C. “Bubonic Plague in Nineteenth-Century China.” Modern China 14 (1988): 107–55.
https://doi.org/10.1177/009770048801400201
Benedictow, O. J. “The Black Death: The Greatest Catastrophe Ever.” History Today 55(3) (2005): 42–49.
______. The Black Death, 1346–1353: The Complete History. Woodbridge: Boydell, 2004.
______. What Disease Was Plague? On the Controversy over the Microbiological Identity of Plague Epidemics of the Past. Leiden: Brill, 2010.
Bianucci, R., L. Rahalison, E. R. Massa, A. Peluso, E. Ferroglio, and M. Signoli. “Technical Note: A Rapid Diagnostic Test Detects Plague in Ancient Human Remains: An Example of the Interaction between Archeological and Biological Approaches (Southeastern France, 16th–18th Centuries).” American Journal of Physical Anthropology 136(3) (2008): 361–67.
https://doi.org/10.1002/ajpa.20818
Bocquet-Appel, J. P., and C. Masset. “Farewell to Paleodemography.” Journal of Human Evolution 11 (1982): 321–33. https://doi.org/10.1016/S0047-2484(82)80023-7
Boldsen, J. L., G. R. Milner, L. W. Konigsberg, and J. W. Wood. “Transition Analysis: A New Method for Estimating Age from Skeletons.” In Paleodemography: Age Distributions from Skeletal Samples, ed. R. D. Hoppa and J. W. Vaupel, 73–106. Cambridge: Cambridge University Press, 2002. https://doi.org/10.1017/CBO9780511542428.005
Bolton, J. L. “‘World Turned Upside Down’: Plague as an Agent of Social and Economic Change.” In The Black Death in England, ed. W. M. Ormrod and P. G. Lindley, 22–46. Stamford: Paul Watkins, 2005.
Bos, K., V. Schuenemann, G. Golding, H. Burbano, N. Waglechner, B. Coombes, J. McPhee, S. DeWitte, M. Myer, S. Schmedes, J. Wood, D. Earn, D. Herring, P. Bauer, H. Poinar, and J. Krause, “A Draft Genome of Yersinia pestis from Victims of the Black Death.” Nature 478 (2011): 506–10. https://doi.org/10.1038/nature10549
Britnell, R. H. “The Black Death in Durham.” Cleveland History: The Bulletin of the Cleveland and Teeside Local History Society 76 (1999): 43–51.
______. “The Black Death in English Towns.” Urban History 21(2) (1994): 195–210.
https://doi.org/10.1017/S0963926800011020
Buikstra, J. E., and L. W. Konigsberg. “Paleodemography: Critiques and Controversies.” American Anthropologist 87 (1985): 316–33. https://doi.org/10.1525/aa.1985.87.2.02a00050
Butcher, A. F. “English Urban Society and the Revolt of 1381.” In The English Rising of 1381, ed. R. H. Hilton and T. H. Aston, 84–111. Cambridge: Cambridge University Press, 2007.
Campbell, B. M. S. The Great Transition: Climate, Disease and Society in the Late Medieval World. The 2013 Ellen McArthur Lecture Series. Cambridge: Cambridge University Press, 2016.https://doi.org/10.1017/CBO9781139031110
______. “Population Pressure, Inheritance, and the Land Market in a Fourteenth-Century Peasant Community.” In Land, Kinship and Lifecycle, ed. R. M. Smith, 87–134. Cambridge, Cambridge University Press, 1984.
Castex, D., and S. Kacki. “Demographic Patterns Distinctive of Epidemic Cemeteries in Archaeological Samples.” Microbiology Spectrum4 (2016).https://doi.org/10.1128/microbiolspec.PoH-0015-2015
Cleri, D. J., and J. R. Vernaleo, L. J. Lombardi, M. S. Rabbat, A. Mathew, R. Marton, M. C. Reyelt. “Plague Pneumonia Disease Caused by Yersinia pestis.” Seminars in Respiratory Infections 12(1) (1997): 12–23.
Cohn, S. K. The Black Death Transformed: Disease and Culture in Early Renaissance Europe. New York: Oxford University Press, 2002.
______. “Epidemiology of the Black Death and Successive Waves of Plague.” Medical History. Supplement 27 (2008): 74–100.
Coulton, G. G. Medieval Panorama: The English Scene from Conquest to Reformation. Cambridge: Cambridge University Press, 1947.
Curtis, D. R., and J. Roosen. “The Sex-selective Impact of the Black Death and Recurring Plagues in the Southern Netherlands, 1349–1450.” Forthcoming in Journal of Physical Anthropology. https://doi.org/10.1002/ajpa.23266
Dean, K. R. “Modeling Plague Transmission in Medieval European Cities.” Master’s Thesis, Department of Biosciences, University of Oslo, 2015 at http:/urn.nb.no/URN:NBN:no-49884
Devault, A. M., K. McLoughlin, C. Jaing, S. Gardner, T. M. Porter, J. M. Enk, J. Thissen, J. Allen, M. Borucki, S. N. DeWitte, A. N. Dhody, and H. N. Poinar. “Ancient Pathogen DNA in Archaeological Samples Detected with a Microbial Detection Array.” Scientific Reports 4 (2014): article #4245. https://doi.org/10.1038/srep04245
DeWitte, S. N. “Age Patterns of Mortality During the Black Death in London, A.D. 1349–1350.” Journal of Archaeological Science 37 (2010): 3394–3400. https://doi.org/10.1016/j.jas.2010.08.006
______. “The Effect of Sex on Risk of Mortality during the Black Death in London, A.D. 1349–1350.” American Journal of Physical Anthropology 139 (2009): 222–34. https://doi.org/10.1002/ajpa.20974
______. “Setting the Stage for Medieval Plague: Pre–Black Death Trends in Survival and Mortality.” American Journal of Physical Anthropology 158 (2015): 441–51. https://doi.org/10.1002/ajpa.22806
DeWitte, S. N., and G. Hughes-Morey. “Stature and Frailty During the Black Death: The Effect of Stature on Risks of Epidemic Mortality in London, A.D. 1348–1350.” Journal of Archaeological Science 39(5) (2012): 1412–19. https://doi.org/10.1016/j.jas.2012.01.019
DeWitte, S. N., and C. M. Stojanowski. “The Osteological Paradox Twenty Years Later: Past Perspectives, Future Directions.” Journal of Archaeological Research 23 (2015): 397–450. https://doi.org/10.1007/s10814-015-9084-1
DeWitte, S. N., and J. W. Wood. “Selectivity of Black Death Mortality with Respect to Preexisting Health.” Proceedings of the National Academy of Sciences of the United States of America 105 (2008): 1436–41. https://doi.org/10.1073/pnas.0705460105
Dickens, B. “Historical Grafitti at Ashwell, Hertfordshire.” In Violet Pritchard, English Medieval Graffiti, Appendix III, 181–83. Cambridge: Cambridge University Press, 2008.
Drali, R., J.-C. Shako, B. Davoust, G. Diatta, and D. Raoult. “A New Clade of African Body and Head Lice Infected by Bartonella Quintana and Yersinia pestis-Democratic Republic of the Congo.” The American Journal of Tropical Medicine and Hygiene 93 (2015): 990–93.
https://doi.org/10.4269/ajtmh.14-0686
Drancourt, M., G. Aboudharam, M. Signoli, O. Dutour, and D. Raoult. “Detection of 400-Year-Old Yersinia pestis DNA in Human Dental Pulp: An Approach to the Diagnosis of Ancient Septicemia.” Proceedings of the National Academy of Science of the United States of America95(21) (1998): 12637–40. https://doi.org/10.1073/pnas.95.21.12637
Drancourt, M., and D. Raoult. “Molecular Insights into the History of Plague.” Microbes and Infection 4(1) (2002): 105–09. https://doi.org/10.1016/S1286-4579(01)01515-5
Ecclestone, M. “Mortality of Rural Landless Men before the Black Death: The Glastonbury Head-tax Lists.” Local Population Studies 63 (1999): 6–29.
Eisen, R. J., S. W. Bearden, A. P. Wilder, J. A. Montenieri, M. F. Antolin, and K. L. Gage. “Early-Phase Transmission of Yersinia pestis by Unblocked Fleas as a Mechanism Explaining Rapidly Spreading Plague Epizootics.” Proceedings of the National Academy of Sciences of the United States of America 103(42) (2006): 15380–85. https://doi.org/10.1073/pnas.0606831103
Fisher, J. L. “The Black Death in Essex.” Essex Review 52 (1943): 13–20.
Gage, T. B. “Are Modern Environments Really Bad for Us? Revisiting the Demographic and Epidemiologic Transitions.” American Journal of Physical Anthropology Supplement 41 (2005): 96–117. https://doi.org/10.1002/ajpa.20353
______. “Bio-Mathematical Approaches to the Study of Human Variation in Mortality.” Yearbook of Physical Anthropology 32 (1989): 185–214. https://doi.org/10.1002/ajpa.1330320509
Gilbert, M. T., J. Cuccui, W. White, N. Lynnerup, R. W. Titball, A. Cooper, and M. B. Prentice. “Absence of Yersinia pestis-Specific DNA in Human Teeth from Five European Excavations of Putative Plague Victims.” Microbiology 150(2) (2004): 341–54. https://doi.org/10.1099/mic.0.26594-0
Gilchrist, R., and B. Sloane. Requiem: The Medieval Monastic Cemetery in Britain. London: Museum of London Archaeology Service, 2005.
Gowland, R. L., and A. T. Chamberlain. “Detecting Plague: Palaeodemographic Characterisation of a Catastrophic Death Assemblage.” Antiquity 79 (2005): 146–57. https://doi.org/10.1017/S0003598X00113766
Grainger, I., and D. Hawkins. “Excavations at the Royal Mint Site 1986–1988.” The London Archaeologist 5 (1988): 429–36.
Grainger, I., D. Hawkins, L. Cowal, and R. Mikulski. The Black Death Cemetery, East Smithfield, London. Museum of London Archaeology Monograph, 43. London: Museum of London Archaeology, 2008.
Grainger, I., and C. Phillpotts. The Cistercian Abbey of St Mary Graces, East Smithfield, London. Museum of London Archaeology Monograph, 44. London: Museum of London Archaeology, 2011.
Green, M. H. “Editor’s Introduction.” The Medieval Globe 1 (2014): 9–26.
______, ed. “Pandemic Disease in the Medieval World: Rethinking the Black Death.” The Medieval Globe 1 (2014).
Groome, N. “The Black Death in the Hundred of Higham Ferrers.” Northamptonshire Past and Present 6 (1982–1983): 309–11.
Gummer, B. The Scourging Angel: The Black Death in the British Isles. London: Vintage, 2009.
Haensch, S., R. Bianucci, M. Signoli, M. Rajerison, M. Schultz, S. Kacki, M. Vermunt, D. A. Weston, D. Hurst, M. Achtman, E. Carniel, and B. Bramanti. “Distinct Clones of Yersinia pestis Caused the Black Death.” PLOS Pathogens 6(10) (2010): e1001134–e34. https://doi.org/10.1371/journal.ppat.1001134
Harvey, P. D. A. A Medieval Oxfordshire Village: Cuxham, 1240–1400. Oxford: Oxford University Press, 1965.
Hatcher, John. Rural Economy and Society in the Duchy of Cornwall, 1390–1500. Cambridge: Cambridge University Press, 1970.
Hawkins, D. “The Black Death and the New London Cemeteries of 1348–1530.” Antiquity 64 (1990): 637–42. https://doi.org/10.1017/S0003598X0007856X
Herlihy, D. The Black Death and the Transformation of the West. Cambridge, Mass.: Harvard University Press, 1997.
Higgins, R. “Emerging or Re-Emerging Bacterial Zoonotic Diseases: Bartonellosis, Leptospirosis, Lyme Borreliosis, Plague.” Revue scientifique et technique 23(2) (2004): 569–81. https://doi.org/10.20506/rst.23.2.1503
Hinckley, A. F., B. J. Biggerstaff, K. S. Griffith, and P. S. Mead. “Transmission Dynamics of Primary Pneumonic Plague in the USA.” Epidemiology and Infection 140(3) (2012): 554–60. https://doi.org/10.1017/S0950268811001245
Hinnebusch, B. J. “Bubonic Plague: A Molecular Genetic Case History of the Emergence of an Infectious Disease.” Journal of Molecular Medicine 75(9) (1997): 645–52.https://doi.org/10.1007/s001090050148
His Majesty’s Stationary Office. 1915. Statistical Abstract Relating to British India from 1903–04 to 1912–13. Forty-eighth Number. London: His Majesty’s Stationary Office, 1915.
Holmes, G. A. “A Decisive Turning Point.” In The Black Death: A Turning Point in History?, ed. W. M. Bowsky, 91–99. New York: Holt, Rhinehart, and Winston, 1971.
Houhamdi, L., H. Lepidi, M. Drancourt, and D. Raoult. “Experimental Model to Evaluate the Human Body Louse as a Vector of Plague.” Journal of Infectious Diseases 194(11) (2006): 1589–96. https://doi.org/10.1086/508995
Hymes, R. “Epilogue: A Hypothesis on the East Asian Beginnings of the Yersinia pestis Polytomy.” The Medieval Globe 1 (2014): 285–308.
James, T. B. The Black Death in Hampshire. Hampshire Papers, 18. Winchester: Hampshire County Council, 1999.
Jonker, M. A. “Estimation of Life Expectancy in the Middle Ages.” Journal of the Royal Statistical Society. Series A (Statistics in Society) 166 (2003): 105–17. https://doi.org/10.1111/1467-985X.00261
Kacki, S., L. Rahalison, M. Rajerison, E. Ferroglio, and R. Bianucci. “Black Death in the Rural Cemetery of Saint-Laurent-De-La-Cabrerisse Aude-Languedoc, Southern France, 14th Century: Immunological Evidence.” Journal of Archaeological Science 38(3) (2011): 581–87. https://doi.org/10.1016/j.jas.2010.10.012
Kowaleski, M. “Gendering Demographic Change.” In The Oxford Handbook of Women and Gender in Medieval Europe, ed. J. M. Bennett and R. M. Karras, 181–96. Oxford: Oxford University Press, 2013.
______. “Medieval People in Town and Country: New Perspectives from Demography and Bioarchaeology.” Speculum 89(3) (2014): 573–600. https://doi.org/10.1017/S0038713414000815
Kreppel, K. S., C. Caminade, S. Telfer, M. Rajerison, L. Rahalison, A. Morse, and M. Baylis. “A Non-Stationary Relationship between Global Climate Phenomena and Human Plague Incidence in Madagascar.” PLoS Neglected Tropical Diseases 8 (2014): e3155. https://doi.org/10.1371/journal.pntd.0003155
Larsen, C. S. Bioarchaeology: Interpreting Behavior from the Human Skeleton, Cambridge Studies in Biological Anthropology, 21. New York: Cambridge University Press, 1997. https://doi.org/10.1017/CBO9780511802676
Lathem, W. W., S. D. Crosby, V. L. Miller, and W. E. Goldman. “Progression of Primary Pneumonic Plague: A Mouse Model of Infection, Pathology, and Bacterial Transcriptional Activity.” Proceedings of the National Academy of Sciences of the United States of America 102(49) (2005): 17786–91. https://doi.org/10.1073/pnas.0506840102
Lewis, C. “Disaster Recovery: New Archaeological Evidence for the Long-Term Impact of the ‘Calamitous’ Fourteenth Century.” Antiquity 90 (2016): 777–97. https://doi.org/10.15184/aqy.2016.69
Lewis, M. E. The Bioarchaeology of Children: Perspectives from Biological and Forensic Anthropology. Cambridge: Cambridge University Press, 2007.
Li, B., C. Du, L. Zhou, Y. Bi, X. Wang, L. Wen, Z. Guo, Z. Song, and R. Yang. “Humoral and Cellular Immune Responses to Yersinia pestisInfection in Long-Term Recovered Plague Patients.” Clinical and Vaccine Immunology 19 (2012): 228–34. https://doi.org/10.1128/CVI.05559-11
Little, L., ed., Plague and the End of Antiquity: The Pandemic of 541–750. Cambridge: Cambridge University Press, 2007.
______. “Plague Historians in Lab Coats.” Past and Present 213 (2011): 267–90. https://doi.org/10.1093/pastj/gtr014
Lock, R. “The Black Death in Walsham-le-Willows.” Proceedings of the Suffolk Institute of Archaeology and History 37 (1992): 316–37.
Lomas, R. A. “The Black Death in County Durham.” Journal of Medieval History 15(2) (1989): 127–40. https://doi.org/10.1016/0304-4181(89)90013-4
Lomas, T. “South-East Durham. Late Fourteenth and Fifteenth Centuries.” In The Peasant Land Market in Medieval England, ed. P. D. A. Harvey, 253–327. Oxford: Oxford University Press, 1984.
McFarlane, K. B. The Nobility of Late Medieval England: The Ford Lectures for 1953 and Related Studies. Oxford: Clarendon Press, 1973.
Megson, B. E. “Morality among London Citizens in the Black Death.” Medieval Prosopography 19 (1998): 125–33.
Migliani, R., S. Chanteau, L. Rahalison, M. Ratsitorahina, J. P. Boutin, L. Ratsifasoamanana, and J. Roux. “Epidemiological Trends for Human Plague in Madagascar During the Second Half of the 20th Century: A Survey of 20,900 Notified Cases.” Tropical Medicine & International Health 11 (2006): 1228–37. https://doi.org/10.1111/j.1365-3156.2006.01677.x
Milner, G. R., and J. L. Boldsen. “Transition Analysis: A Validation Study with Known-Age Modern American Skeletons.” American Journal of Physical Anthropology 148 (2012): 98–110. https://doi.org/10.1002/ajpa.22047
Milner, G. R., J. W. Wood, and J. L. Boldsen. “Paleodemography.” In Biological Anthropology of the Human Skeleton, ed. M. Katzenberg and S. Saunders, 561–600. New York: Wiley-Liss, 2008. https://doi.org/10.1002/9780470245842.ch18
Moore, S. M., A. Monaghan, J. N. Borchert, J. T. Mpanga, L. A. Atiku, K. A. Boegler, J. Montenieri, K. MacMillan, K. L. Gage, and R. J. Eisen. “Seasonal Fluctuations of Small Mammal and Flea Communities in a Ugandan Plague Focus: Evidence to Implicate arvicanthis niloticus and Crocidura spp. as Key Hosts in Yersinia pestis Transmission.” Parasites & Vectors 8 (2015): 11. https://doi.org/10.1186/s13071-014-0616-1
Mullan, J. “Mortality, Gender and the Plague of 1361–2 on the Estate of the Bishop of Winchester.” Cardiff Historical Papers, 2007/8 (2007): 1–41.
Müller, H. G., and B. Love, R. D. Hoppa. “Semiparametric Method for Estimating Paleodemographic Profiles from Age Indicator Data.” American Journal of Physical Anthropology 117(1) (2002): 1–14. https://doi.org/10.1002/ajpa.10000
Nash, A. E. “The Mortality Pattern of the Wiltshire Lords of the Manor, 1242–1377.” Southern History 2 (1980): 31–43.
Nishiura, H. “Epidemiology of a Primary Pneumonic Plague in Kantoshu, Manchuria, from 1910 to 1911: Statistical Analysis of Individual Records Collected by the Japanese Empire.” International Journal of Epidemiology 35 (2006): 1059–65. https://doi.org/10.1093/ije/dyl091
Ohlin, G. “No Safety in Numbers: Some Pitfalls of Historical Statistics.” In Industrialization in Two Systems: Essays in Honor of Alexander Gerschenkron, 68–90. New York: John Wiley and Sons, 1966.
Olea, R. A. and G. Christakos. “Duration of Urban Mortality for the Fourteenth-Century Black Death Epidemic.” Human Biology 77(3) (2005): 291–303. https://doi.org/10.1353/hub.2005.0051
Ortner, D. J. Identification of Pathological Conditions in Human Skeletal Remains, 2nd edition. San Diego: Academic Press, 2003.
______. “Theoretical and Methodological Iissues in Paleopathology.” In Human Paleopathology: Current Syntheses and Future Options, ed. D. J. Ortner and A. C. Aufderheide, 5–11. Washington, D.C.: Smithsonian Institution Press, 1991.
Page, F. M. The Estates of Crowland Abbey: A Study in Manorial Organization. Cambridge: Cambridge University Press, 1934.
Pechous, R. D., V. Sivaraman, N. M. Stasulli, and W. E. Goldman. “Pneumonic Plague: The Darker Side of Yersinia pestis.” Trends in Microbiology 24 (2016): 190–97. https://doi.org/10.1016/j.tim.2015.11.008
Perry, M. A. “Is Bioarchaeology a Handmaiden to History? Developing a Historical Bioarchaeology.” Journal of Anthropological Archaeology 26(3) (2007): 486–515. https://doi.org/10.1016/j.jaa.2007.02.002
Perry, R. D., and J. D. Fetherston. “Yersinia pestis: Etiologic Agent of Plague.” Clinical Microbiology Reviews 10(1) (1997): 35–66.
Plague Research Commission. “The Epidemiological Observations Made by the Commission in Bombay City.” Journal of Hygiene 7 (1907): 724–98. https://doi.org/10.1017/S0022172400033684
Poos, L. R. A Rural Society after the Black Death: Essex, 1350–1525. Cambridge: Cambridge University Press, 1991. https://doi.org/10.1017/CBO9780511522437
Poos, L. R., J. E. Oeppen, and R. M. Smith. “Re-assessing Josiah Russell’s Measurements of Late Medieval Mortality Using the Inquisitions Post Mortem.” In The Fifteenth-Century Inquisitions Post Mortem: A Companion, ed. M. Hicks, 155–68. Woodbridge: Boydell, 2012.
Postan, M. M., and J. Z. Titow. “Heriots and Prices on Winchester Manors.” Economic History Review, 2nd series, 11 (1958–1959): 392–415.
Postles, D. “Demographic Change in Kibworth Harcourt, Leicestershire in the Later Middle Ages.” Local Population Studies 8 (1992): 41–48.
Pusch, C. M., L. Rahalison, N. Blin, G. J. Nicholson, and A. Czarnetzki. “Yersinial F1 Antigen and the Cause of Black Death.” The Lancet Infectious Diseases 4(8) (2004): 484–85. https://doi.org/10.1016/S1473-3099(04)01099-0
Raoult, D., G. Aboudharam, E. Crubezy, G. Larrouy, B. Ludes, and M. Drancourt. “Molecular Identification by ‘Suicide PCR’ of Yersinia pestis as the Agent of Medieval Black Death.” Proceedings of the National Academy of Sciences in the United States of America 97 (2000): 12800–03. https://doi.org/10.1073/pnas.220225197
Ratovonjato, J., M. Rajerison, S. Rahelinirina, and S. Boyer. “Yersinia pestis in Pulex Irritans Fleas During Plague Outbreak, Madagascar.” Emerging Infectious Diseases 20 (2014): 1414–15. https://doi.org/10.3201/eid2008.130629
Ravensdale, J. “Population Changes and Transfer of Customary Land on a Cambridgeshire Manor in the I4th Century.” In Land, Kinship and Lifecycle, ed. R. M. Smith, 197–225. Cambridge: Cambridge University Press, 1984.
Razi, Z. Life, Marriage and Death in a Medieval Parish: Economy, Society and Demography in Halesowen, 1270–1400. Cambridge: Cambridge University Press, 1980.
Richard, V., J. M. Riehm, P. Herindrainy, R. Soanandrasana, M. Ratsitoharina, F. Rakotomanana, S. Andrianalimanana, H. C Scholz, and M. Rajerison. “Pneumonic Plague Outbreak, Northern Madagascar, 2011.” Emerging Infectious Disease Journal 21 (2015): 8–15. https://doi.org/10.3201/eid2101.131828
Rogers, J., and T. Waldron. “DISH and the Monastic Way of Life.” International Journal of Osteoarchaeology 11 (2001): 357–65. https://doi.org/10.1002/oa.574
Röhrkasten, J. “Trends of Mortality in Late Medieval London (1348–1400).” Nottingham Medieval Studies 45 (2001): 172–209. https://doi.org/10.1484/J.NMS.3.326
Royer, K. “The Blind Men and the Elephant: Imperial Medicine, Medieval Historians, and the Role of Rats in the Historiography of Plague.” In Medicine and Colonialism: Historical Perspectives in India and South Africa, ed. Bala Poonam, 99–110. London: Pickering and Chatto, 2014.
Rubini, M., E. Gualdi-Russo, V. S. Manzon, N. Rinaldo, and R. Bianucci. “Mortality Risk Factors Show Similar Trends in Modern and Historic Populations Exposed to Plague.” Journal of Infection in Developing Countries 10 (2016): 488–93. https://doi.org/10.3855/jidc.7974
Russell, J. C. British Medieval Population. Albuquerque: University of New Mexico Press, 1948.
Sattenspiel, L., and H. Harpending. “Stable Populations and Skeletal Age.” American Antiquity 48(3) (1983): 489–98. https://doi.org/10.2307/280557
Schuenemann, V. J., S. Bos, S. Dewitte, S. Schmedes, J. Jamieson, A. Mittnik, S. Forrest, B. K. Coombes, J. W. Wood, D. J. D. Earn, W. White, J. Krause, and H. N. Poinar. “Targeted Enrichment of Ancient Pathogens Yielding the Ppcp1 Plasmid of Yersinia pestis from Victims of the Black Death.” Proceedings of the National Academy of Sciences of the United States of America 108 (2011): e746–e52. https://doi.org/10.1073/pnas.1105107108
Scott, S., and C. J. Duncan. Biology of Plagues: Evidence from Historical Populations. Cambridge: Cambridge University Press, 2001. https://doi.org/10.1017/CBO9780511542527
Seifert, L., I. Wiechmann, M. Harbeck, A. Thomas, G. Grupe, M. Projahn, H. C. Scholz, and J. M. Riehm. “Genotyping Yersinia pestis in Historical Plague: Evidence for Long-Term Persistence of Y. pestis in Europe from the 14th to the 17th Century.” PloS One 11 (2016): e0145194. https://doi.org/10.1371/journal.pone.0145194
Shrewsbury, J. F. D. Bubonic Plague in the British Isles. Cambridge: Cambridge University Press, 1970.
Sloane, B. The Black Death in London. Stroud: The History Press, 2011.
Thompson, M. G. “The Polden Hill Manors of Glastonbury Abbey: Land and People circa 1260 to 1351.” Unpublished Ph.D. thesis, University of Leicester, 1997.
Titow, J. Z. English Rural Society, 1200–1350. London: George Allen and Unwin, 1969.
Tran, T.-N.-N., C. L. Forestier, M. Drancourt, D. Raoult, and G. Aboudharam. “Brief Communication: Co-Detection of Bartonella Quintana and Yersinia pestis in an 11th–15th Burial Site in Bondy, France.” American Journal of Physical Anthropology 145(3) (2011): 489–94. https://doi.org/10.1002/ajpa.21510
Twigg, G. The Black Death: A Biological Reappraisal. London: Batsford, 1984.
______. “The Black Death and DNA.” The Lancet Infectious Diseases 3(1) (2003): 11.
Usher, B. M. “A Multistate Model of Health and Mortality for Paleodemography: Tirup Cemetery.” PhD dissertation, Pennsylvania State University, 2000.
Van Gerven, D. P., and G. J. Armelagos. “‘Farewell to Paleodemography’? Rumors of Its Death Have Been Greatly Exaggerated.” Journal of Human Evolution 12(4) (1983): 353–60. https://doi.org/10.1016/S0047-2484(83)80162-6
Vaupel, J. W., K. G. Manton, and E. Stallard. “The Impact of Heterogeneity in Individual Frailty on the Dynamics of Mortality.” Demography 16 (1979): 439–54. https://doi.org/10.2307/2061224
Wagner, D. M., J. Klunk, M. Harbeck, A. Devault, N. Waglechner, J. W. Sahl, J. Enk, D. N. Birdsell, M. Kuch, C. Lumibao, D. Poinar, T. Pearson, M. Fourment, B. Golding, J. M. Riehm, D. J. D. Earn, S. DeWitte, J.-M. Rouillard, G. Grupe, I. Wiechmann, J. B. Bliska, P. S. Keim, H. C. Scholz, E. C. Holmes, and H. Poinar. “Yersinia pestis and the Plague of Justinian 541–543 AD: A Genomic Analysis.” The Lancet Infectious Diseases 14(4) (2014): 319–26. https://doi.org/10.1016/S1473-3099(13)70323-2
Watts, D. G. “The Black Death in Dorset and Hampshire.” In “The Black Death in Wessex,” ed. T. B. James. The Hatcher Review 5(46) (1998): 21–28.
Weston, D. A. “Investigating the Specificity of Periosteal Reactions in Pathology Museum Specimens.” American Journal of Physical Anthropology 137(1) (2008): 48–59. https://doi.org/10.1002/ajpa.20839
Wiechmann, I., and G. Grupe. “Detection of Yersinia pestis DNA in Two Early Medieval Skeletal Finds from Aschheim (Upper Bavaria, 6th Century A.D.).” American Journal of Physical Anthropology 126(1) (2005): 48–55. https://doi.org/10.1002/ajpa.10276
Wood, J. W., and S. N. DeWitte-Aviña. “Was the Black Death Yersinial Plague?” The Lancet Infectious Diseases 3 (2003): 327–28. https://doi.org/10.1016/S1473-3099(03)00651-0
Wood, J. W., R. J. Ferrell, and S. N. DeWitte-Aviña. “The Temporal Dynamics of the Fourteenth-Century Black Death: New Evidence from English Ecclesiastical Records.” Human Biology 75(4) (2003): 427–48. https://doi.org/10.1353/hub.2003.0067
Wood, J. W., D. J. Holman, K. A. O’Connor, and R. J. Ferrell. “Mortality Models for Paleodemography.” In Paleodemography: Age Distributions from Skeletal Samples, ed. R. D. Hoppa and J. W. Vaupel, 129–68. Cambridge: Cambridge University Press, 2002. https://doi.org/10.1017/CBO9780511542428.007
Wood, J. W., G. R. Milner, H. C. Harpending, and K. M. Weiss. “The Osteological Paradox: Problems of Inferring Prehistoric Health from Skeletal Samples.” Current Anthropology 33 (1992): 343–70. https://doi.org/10.1086/204084
Ziegler, M. “The Black Death and the Future of the Plague.” The Medieval Globe 1 (2014): 259–84.
______. “The Case for Louse-Transmitted Plague.” Available at https://contagions.wordpress.com/2016/11/17/the-case-for-louse-transmitted-plague/ (accessed 23 January 2017).
Ziegler, Philip. The Black Death. Harmondsworth: Penguin Books, 1982. First published by Collins, 1969.
Originally published by the Fragments, Vol. 6 (2017), under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported license.