Researchers have identified the role that a four-stranded version of DNA may play in the role of cancer progression, and suggest that it may be used to develop new targeted cancer therapies.
09.12.2016
Scientists have identified where a four-stranded version of DNA exists within the genome of human cells, and suggest that it may hold a key to developing new, targeted therapies for cancer.
In work funded by Cancer Research UK and EMBO, the researchers, from the University of Cambridge, found that these quadruple helix structures occur in the regions of DNA that control genes, particularly cancer genes, suggesting that they may play a role in switching genes on or off. The results, reported in the journal Nature Genetics, could also have implications for cancer diagnostics and the development of new targeted treatments.
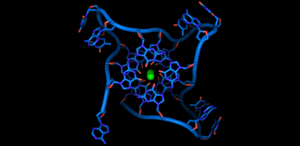
Most of us are familiar with the double helix structure of DNA, but there is also a version of the molecule which has a quadruple helix structure. These structures are often referred to as G-quadruplexes, as they form in the regions of DNA that are rich in the building block guanine, usually abbreviated to ‘G’. These structures were first found to exist in human cells by the same team behind the current research, but at the time it was not exactly clear where these structures were found in the genome, and what their role was, although it was suspected that they had a link with certain cancer genes.
“There have been a number of different connections made between these structures and cancer, but these have been largely hypothetical,” said Professor Shankar Balasubramanian, from Cambridge’s Department of Chemistry and Cancer Research UK Cambridge Institute, and the paper’s senior author. “But what we’ve found is that even in non-cancer cells, these structures seem to come and go in a way that’s linked to genes being switched on or off.”
Starting with a pre-cancerous human cell line, the researchers used small molecules to change the state of the cells in order to observe where the G-quadruplexes might appear. They detected approximately 10,000 G-quadruplexes, primarily in regions of DNA associated with switching genes on or off, and particularly in genes associated with cancer.
“What we observed is that the presence of G-quadruplexes goes hand in hand with the output of the associated gene,” said Balasubramanian. This suggests that G-quadruplexes may play a similar role to epigenetic marks: small chemical modifications which affect how the DNA sequence is interpreted and control how certain genes are switched on or off.
The results also suggest that G-quadruplexes hold potential as a molecular target for early cancer diagnosis and treatment, in particular for so-called small molecule treatments which target cancer cells, instead of traditional treatments which hit all cells.
“We’ve been looking for an explanation for why it is that certain cancer cells are more sensitive to small molecules that target G-quadruplexes than non-cancer cells,” said Balasubramanian. “One simple reason could be that there are more of these G-quadruplex structures in pre-cancerous or cancer cells, so there are more targets for small molecules, and so the cancer cells tend to be more sensitive to this sort of intervention than non-cancer cells.
“It all points in a certain direction, and suggests that there’s a rationale for the selective targeting of cancer cells.”
“We found that G-quadruplexes appear in regions of the genome where proteins such as transcription factors control cell fate and function,” said Dr Robert Hänsel-Hertsch, the paper’s lead author. “The finding that these structures may help regulate the way that information is encoded and decoded in the genome will change the way we think this process works.”
Dr Emma Smith, Cancer Research UK’s science information manager, said: “Figuring out the fundamental processes that cancer cells use to switch genes on and off could help scientists develop new treatments that work against many types of the disease. And exploiting weaknesses in cancer cells could mean this approach would cause less damage to healthy cells, reducing potential side effects. It’s still early days, but promising leads like this are where the treatments of the future will come from.”
Reference:
Robert Hänsel-Hertsch et. al. ‘G-quadruplex structures mark human regulatory chromatin.’ Nature Genetics (2016). DOI: 10.1038/ng.3662